This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
User:David Canner/Sandbox good
From Proteopedia
How to make excellent scenes:
This is a list of tips and tricks to develop effective scenes for your pages.
Tip #1: When developing a series of scenes illustrating related parts of a protein, use the “transition options” to create smooth transitions void of peculiar zoom-outs, etc.
Scene Transitions
Smooth Transitions
| |||||||||||
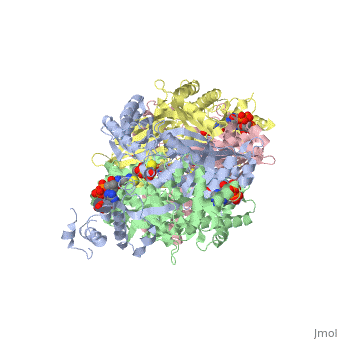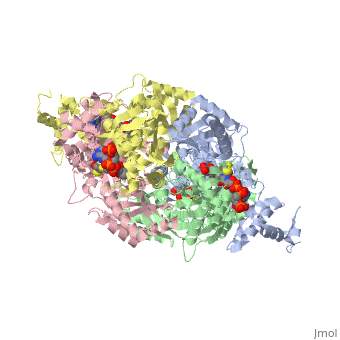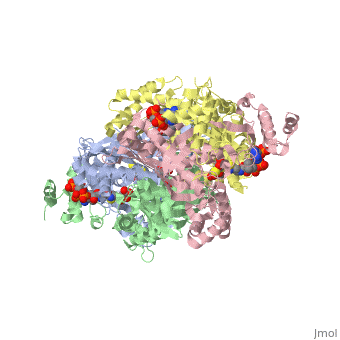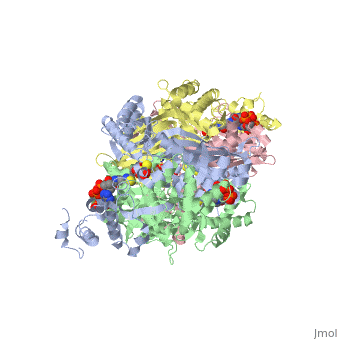
Example from the page HMG-CoA Reductase:
The HMG binding pocket is the site of catalysis in HMGR. is a critical structural element of this binding site. Residues and are positioned in the active site as is . It is this K691 that likely stabilizes the negatively charged oxygen of the first mevaldyl-CoA intermediate. The mevaldyl CoA intermediate is subsequently converted to Mavaldehyde with added stabilization from . It is then believed that the close proximity of increases the pKA of E559, allowing it to be a proton donor for the reduction of mevaldehyde into mevalonate.
Compared with: The HMG binding pocket is the site of catalysis in HMGR. is a critical structural element of this binding site. Residues and are positioned in the active site as is . Etc…