This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Sandbox Reserved 195
From Proteopedia
| This Sandbox is Reserved from Feb 02, 2011, through Jul 31, 2011 for use by the Biochemistry II class at the Butler University at Indianapolis, IN USA taught by R. Jeremy Johnson. This reservation includes Sandbox Reserved 191 through Sandbox Reserved 200. |
To get started:
More help: Help:Editing |
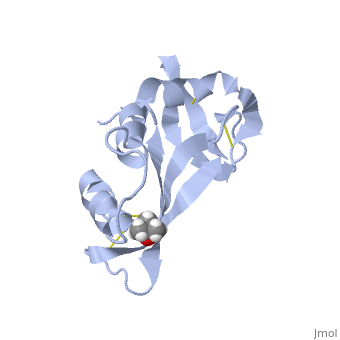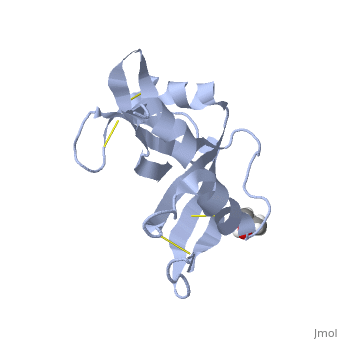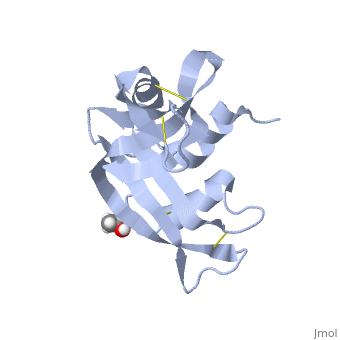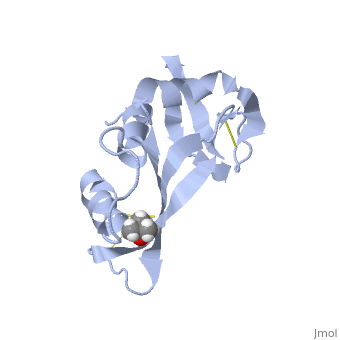
Ribonuclease S
Ribonuclease S consists of two fragments of bovine Ribonuclease A, S peptide and S protein which make up the peptide-protein complex. RNase S has been studied to reveal mechanisms of protein folding by coupling folding and association. Studies of RNase S lead to greater interest in the investigation of RNase A and its role in biological systems. Through such studies, it has been suggested that RNase A and its oligomers could have anti-cancer effects, and a greater knowledge of the correlation between protein folding and enzyme activity.
|
RNase S is RNase A treated with subtilisin, which cleaves a single peptide bond. The on RNase A is located between residues Ala 20 and Ser 21. Upon cleavage the fragments S peptide and S protein are identified giving RNase S its structure. consists of residues 1-20, and is largely responsible for proper folding. S protein is comprised of residues 21-124.
The of RNase S consists of residues His 12, Lys 41, Val 43, Asn 44, Thr 45, His 119, Phe 120, Asp 121, and Ser 123.
Water, Hydrophobic Effect, Prolines
is conserved between RNase
|