This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
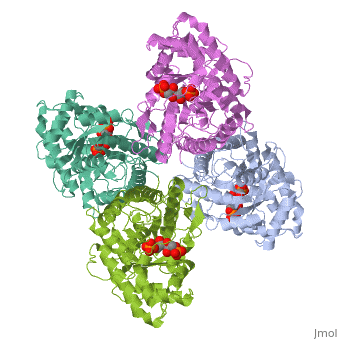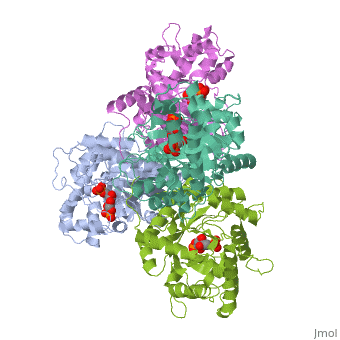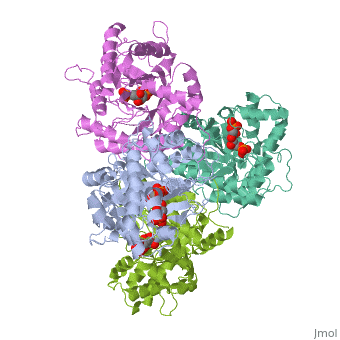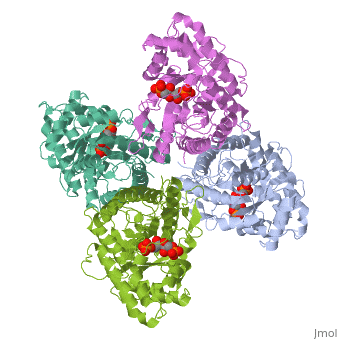
4ald
From Proteopedia
| |||||||||
| 4ald, resolution 2.80Å () | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Ligands: | |||||||||
| Activity: | Fructose-bisphosphate aldolase, with EC number 4.1.2.13 | ||||||||
| |||||||||
| |||||||||
| Resources: | FirstGlance, OCA, RCSB, PDBsum | ||||||||
| Coordinates: | save as pdb, mmCIF, xml | ||||||||
Contents |
HUMAN MUSCLE FRUCTOSE 1,6-BISPHOSPHATE ALDOLASE COMPLEXED WITH FRUCTOSE 1,6-BISPHOSPHATE
Template:ABSTRACT PUBMED 10048322
Disease
[ALDOA_HUMAN] Defects in ALDOA are the cause of glycogen storage disease type 12 (GSD12) [MIM:611881]; also known as red cell aldolase deficiency. A metabolic disorder associated with increased hepatic glycogen and hemolytic anemia. It may lead to myopathy with exercise intolerance and rhabdomyolysis.[1][2][3][4][5]
Function
[ALDOA_HUMAN] Plays a key role in glycolysis and gluconeogenesis. In addition, may also function as scaffolding protein (By similarity).
About this Structure
4ald is a 1 chain structure with sequence from Homo sapiens. The February 2004 RCSB PDB Molecule of the Month feature on The Glycolytic Enzymes by David S. Goodsell is 10.2210/rcsb_pdb/mom_2004_2. Full crystallographic information is available from OCA.
See Also
Reference
- Dalby A, Dauter Z, Littlechild JA. Crystal structure of human muscle aldolase complexed with fructose 1,6-bisphosphate: mechanistic implications. Protein Sci. 1999 Feb;8(2):291-7. PMID:10048322
- ↑ Esposito G, Vitagliano L, Costanzo P, Borrelli L, Barone R, Pavone L, Izzo P, Zagari A, Salvatore F. Human aldolase A natural mutants: relationship between flexibility of the C-terminal region and enzyme function. Biochem J. 2004 May 15;380(Pt 1):51-6. PMID:14766013 doi:10.1042/BJ20031941
- ↑ Kishi H, Mukai T, Hirono A, Fujii H, Miwa S, Hori K. Human aldolase A deficiency associated with a hemolytic anemia: thermolabile aldolase due to a single base mutation. Proc Natl Acad Sci U S A. 1987 Dec;84(23):8623-7. PMID:2825199
- ↑ Takasaki Y, Takahashi I, Mukai T, Hori K. Human aldolase A of a hemolytic anemia patient with Asp-128----Gly substitution: characteristics of an enzyme generated in E. coli transfected with the expression plasmid pHAAD128G. J Biochem. 1990 Aug;108(2):153-7. PMID:2229018
- ↑ Kreuder J, Borkhardt A, Repp R, Pekrun A, Gottsche B, Gottschalk U, Reichmann H, Schachenmayr W, Schlegel K, Lampert F. Brief report: inherited metabolic myopathy and hemolysis due to a mutation in aldolase A. N Engl J Med. 1996 Apr 25;334(17):1100-4. PMID:8598869 doi:http://dx.doi.org/10.1056/NEJM199604253341705
- ↑ Yao DC, Tolan DR, Murray MF, Harris DJ, Darras BT, Geva A, Neufeld EJ. Hemolytic anemia and severe rhabdomyolysis caused by compound heterozygous mutations of the gene for erythrocyte/muscle isozyme of aldolase, ALDOA(Arg303X/Cys338Tyr). Blood. 2004 Mar 15;103(6):2401-3. Epub 2003 Nov 13. PMID:14615364 doi:10.1182/blood-2003-09-3160