This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Sandbox Reserved 687
From Proteopedia
| This Sandbox is Reserved from 30/01/2013, through 30/12/2013 for use in the course "Biochemistry II" taught by Hannah Tims at the Messiah College. This reservation includes Sandbox Reserved 686 through Sandbox Reserved 700. |
To get started:
More help: Help:Editing |
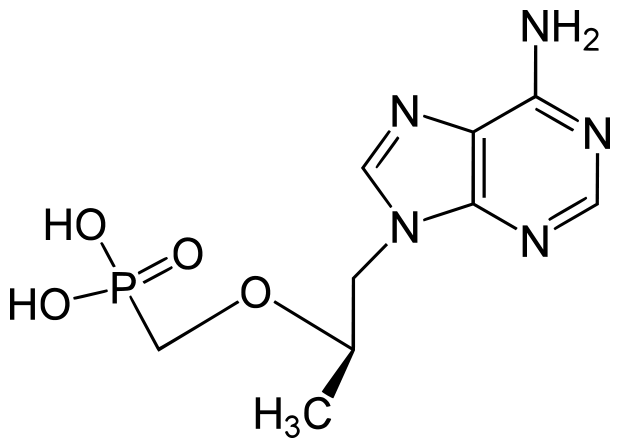
Complera: Emtricitabine/rilpivirine/tenofovir
- Marketed by: Gilead Sciences and Janssen Therapeutics
- Major Indication: HIV infection
- Drug Class: Combination of nucleoside/tide reverse transcriptase inhibitor and nonnucleoside reverse transcriptase inhibitor
- Date of FDA Approval: August 2011
Rilpivirine (RPV)
|
RPV in the In red are the binding pocket residues p66 (Leu-100, Lys-101, Lys-103, Val-106, Thr-107, Val-108, Val-179, Tyr-181, Tyr-188, Val-189, Gly-190, Phe-227, Trp-229, Leu- 234, and Tyr-318) and p51(Glu-138). Singh et al.
|
Reverse Transcriptase:
Emtricitabine Image:Emtricitabine3.png
|