Sandbox Reserved 19
From Proteopedia
| This Sandbox is Reserved from May 10, 2015, through July 31, 2015 for use by the class Protein 3D Structure Visualization & Structural Bioinformatics taught by Eric Martz and Keiichi Namba at Osaka University, Japan. This reservation includes Sandbox Reserved 1 through Sandbox Reserved 10. Syllabus. |
To get started:
More help: Getting Started in Proteopedia, Help:Editing, Main Help Page. |
|
Cationic+ atoms are colored blue. 65+, including 25+ with incomplete sidechains. (21 Arg, 44 Lys) (6 His) Anionic- atoms are colored red. 47-, including 12- with incomplete sidechains. (22 Asp, 24 Glu, 1 C terminus)
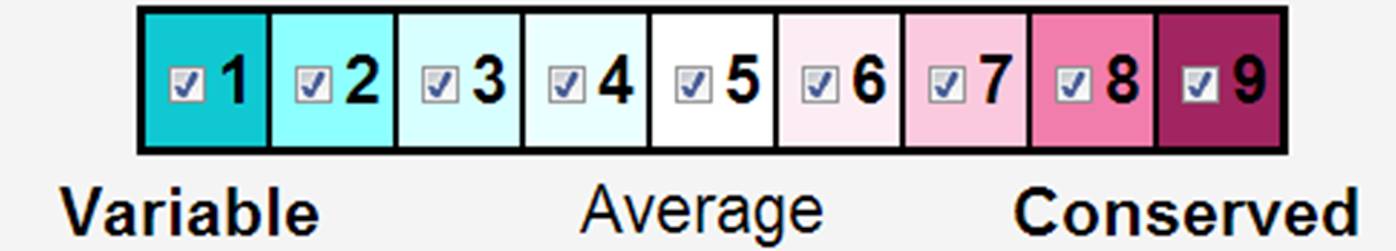
The catalytic pocket is highly conserved, while conservation of the remainder of the surface is more variable. Conserved regions of unknown function deserve investigation because they are likely to have important functions.