This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
1qrq
From Proteopedia
| |||||||
| , resolution 2.80Å | |||||||
|---|---|---|---|---|---|---|---|
| Ligands: | |||||||
| Coordinates: | save as pdb, mmCIF, xml | ||||||
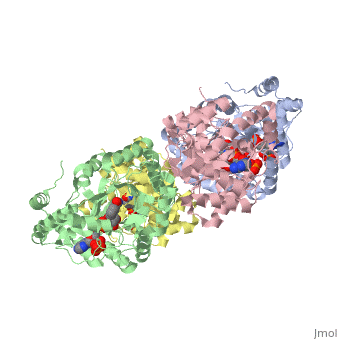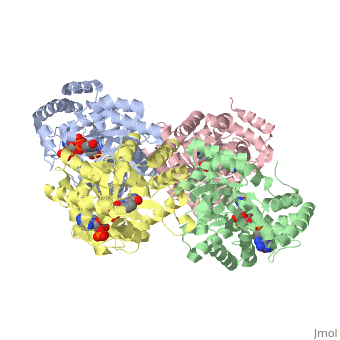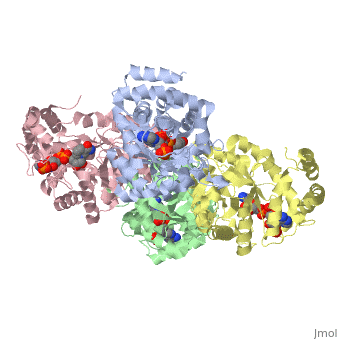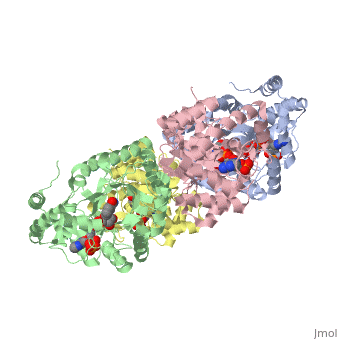
STRUCTURE OF A VOLTAGE-DEPENDENT K+ CHANNEL BETA SUBUNIT
Overview
The integral membrane subunits of many voltage-dependent potassium channels are associated with an additional protein known as the beta subunit. One function of beta subunits is to modify K+ channel gating. We have determined the structure of the conserved core of mammalian beta subunits by X-ray crystallography at 2.8 A resolution. Like the integral membrane component of K+ channels, beta subunits form a four-fold symmetric structure. Each subunit is an oxidoreductase enzyme complete with a nicotinamide co-factor in its active site. Several structural features of the enzyme active site, including its location with respect to the four-fold axis, imply that it may interact directly or indirectly with the K+ channel's voltage sensor. This structure suggests a mechanism for coupling membrane electrical excitability directly to chemistry of the cell.
About this Structure
1QRQ is a Single protein structure of sequence from Rattus norvegicus. Full crystallographic information is available from OCA.
Reference
Structure of a voltage-dependent K+ channel beta subunit., Gulbis JM, Mann S, MacKinnon R, Cell. 1999 Jun 25;97(7):943-52. PMID:10399921
Page seeded by OCA on Thu Mar 20 13:41:52 2008