This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
1ygp
From Proteopedia
| |||||||
| , resolution 2.8Å | |||||||
|---|---|---|---|---|---|---|---|
| Ligands: | and | ||||||
| Gene: | YEAST GLYCOGEN PHOSPHORYLASE (Saccharomyces cerevisiae) | ||||||
| Coordinates: | save as pdb, mmCIF, xml | ||||||
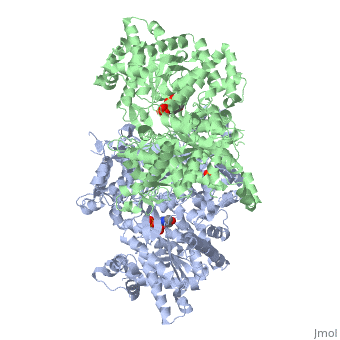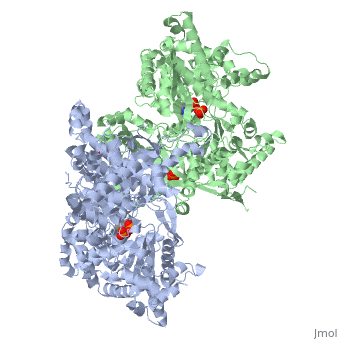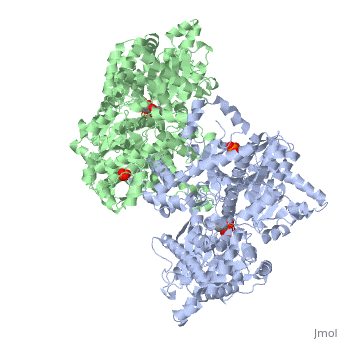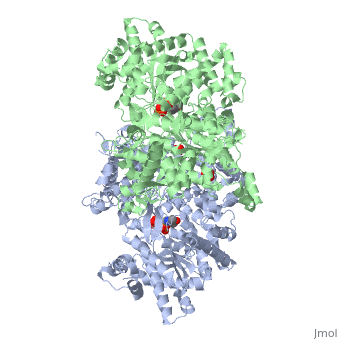
PHOSPHORYLATED FORM OF YEAST GLYCOGEN PHOSPHORYLASE WITH PHOSPHATE BOUND IN THE ACTIVE SITE.
Overview
A phosphorylation-initiated mechanism of local protein refolding activates yeast glycogen phosphorylase (GP). Refolding of the phosphorylated amino-terminus was shown to create a hydrophobic cluster that wedges into the subunit interface of the enzyme to trigger activation. The phosphorylated threonine is buried in the allosteric site. The mechanism implicates glucose 6-phosphate, the allosteric inhibitor, in facilitating dephosphorylation by dislodging the buried covalent phosphate through binding competition. Thus, protein phosphorylation-dephosphorylation may also be controlled through regulation of the accessibility of the phosphorylation site to kinases and phosphatases. In mammalian glycogen phosphorylase, phosphorylation occurs at a distinct locus. The corresponding allosteric site binds a ligand activator, adenosine monophosphate, which triggers activation by a mechanism analogous to that of phosphorylation in the yeast enzyme.
About this Structure
1YGP is a Single protein structure of sequence from Saccharomyces cerevisiae. The following page contains interesting information on the relation of 1YGP with [Glycogen Phosphorylase]. Full crystallographic information is available from OCA.
Reference
A protein phosphorylation switch at the conserved allosteric site in GP., Lin K, Rath VL, Dai SC, Fletterick RJ, Hwang PK, Science. 1996 Sep 13;273(5281):1539-42. PMID:8703213
Page seeded by OCA on Thu Mar 20 15:22:42 2008