This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
1js3
From Proteopedia
| |||||||
| , resolution 2.25Å | |||||||
|---|---|---|---|---|---|---|---|
| Ligands: | , and | ||||||
| Activity: | Aromatic-L-amino-acid decarboxylase, with EC number 4.1.1.28 | ||||||
| Coordinates: | save as pdb, mmCIF, xml | ||||||
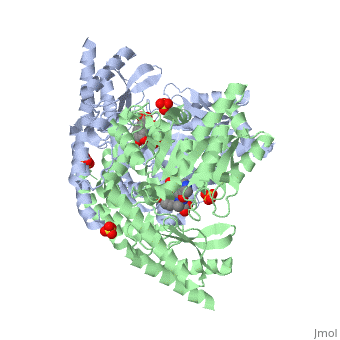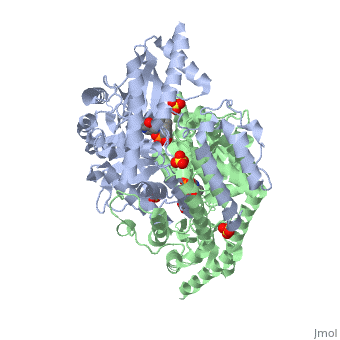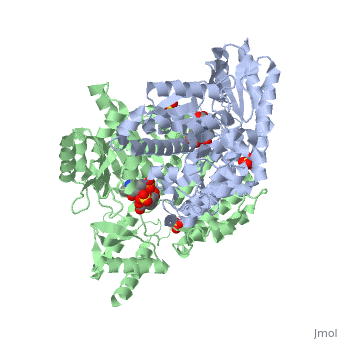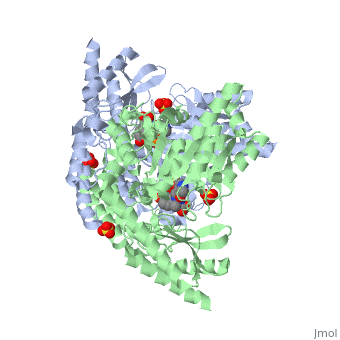
Crystal structure of dopa decarboxylase in complex with the inhibitor carbidopa
Overview
DOPA decarboxylase (DDC) is responsible for the synthesis of the key neurotransmitters dopamine and serotonin via decarboxylation of L-3,4-dihydroxyphenylalanine (L-DOPA) and L-5-hydroxytryptophan, respectively. DDC has been implicated in a number of clinic disorders, including Parkinson's disease and hypertension. Peripheral inhibitors of DDC are currently used to treat these diseases. We present the crystal structures of ligand-free DDC and its complex with the anti-Parkinson drug carbiDOPA. The inhibitor is bound to the enzyme by forming a hydrazone linkage with the cofactor, and its catechol ring is deeply buried in the active site cleft. The structures provide the molecular basis for the development of new inhibitors of DDC with better pharmacological characteristics.
About this Structure
1JS3 is a Single protein structure of sequence from Sus scrofa. Full crystallographic information is available from OCA.
Reference
Structural insight into Parkinson's disease treatment from drug-inhibited DOPA decarboxylase., Burkhard P, Dominici P, Borri-Voltattorni C, Jansonius JN, Malashkevich VN, Nat Struct Biol. 2001 Nov;8(11):963-7. PMID:11685243
Page seeded by OCA on Sun Mar 23 12:23:25 2008