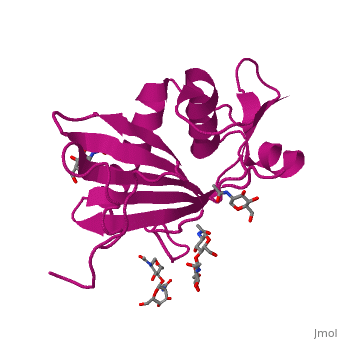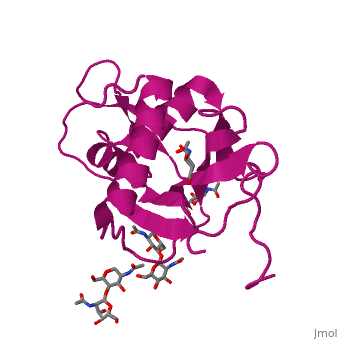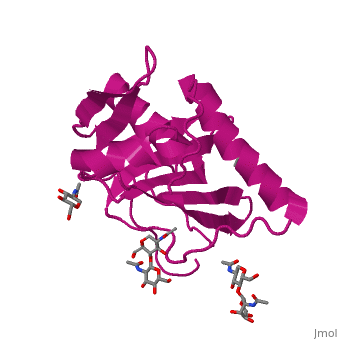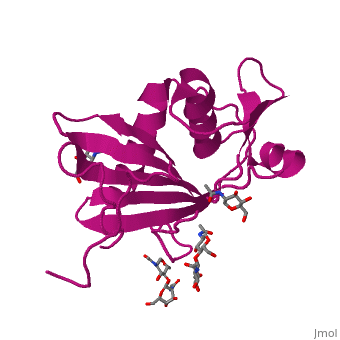Importance
Lassa virus (LASV), an Old World arenavirus, is a notorious disease-causing agent primarily in West Africa that is able to spread to rodents, as well as humans. This deadly pathogen causes severe viral hemorrhagic fevers and significant mortality. So far, there are no available vaccines for LASV or any other viruses found in the Arenaviridae family. Determining the structure of the complete trimeric glycoprotein complex (GPC), composed of GP1, GP2, and SSP (stable signal peptide), will lay the foundation for a future discovery of novel antiviral drugs. This is the first representative structure for Old World arenaviruses.
Function
GP1 (Glycoprotein 1) is the receptor binding domain of LASV that mediates receptor recognition. Research thus far indicates that GP1 from LASV may undergo irreversible conformational changes that could serve as an immunological decoy mechanism.
Structural Highlights
GP1 of LASV is a 4 chain structure with ligands attached to each chain. The overall architecture of GP1 features a central β-sheet and two distinct halves: a glycosylated half containing the receptor-binding site that is made mostly by the central β-sheet and surrounding loops and a half that contains mostly helices and most likely faces the trimer axis[1]. The method used to determine this structure was X-ray diffraction
Histidine Triad
Attached to this structure is a unique that is highly conserved among Old World arenaviruses. Located on the β-sheet face of GP1, the histidine triad is a structural element that directly interacts with LAMP1 (Lysosome-associated membrane glycoprotein) and helps stabilize a LAMP1-"compatible" conformation by providing a molecular mechanism for the pH-dependent receptor switching[1]. The is critical in forming a for LAMP1.
LAMP1 Binding Site
The primary cellular receptor of LASV α-dystroglycan (α-DG), which is recognized by a trimeric class 1 viral GPC on the surface of the virus. Following successful attachment to α-DG on cells, LASV is internalized via macropinocytosis, and the GPC facilitates membrane fusion at the acidic environment of a late endosomal compartment. Recent studies have shown that successful infection by LASV requires it to switch in a pH-dependent manner from α-DG to LAMP1.
Resources
For more information on this protein structure visit the following sites: FirstGlance, OCA, PDBe, RCSB, PDBsum, ProSAT