This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Sandbox Reserved 1347
From Proteopedia
| This Sandbox is Reserved from January through July 31, 2018 for use in the course HLSC322: Principles of Genetics and Genomics taught by Genevieve Houston-Ludlam at the University of Maryland, College Park, USA. This reservation includes Sandbox Reserved 1311 through Sandbox Reserved 1430. |
To get started:
More help: Help:Editing |
Contents |
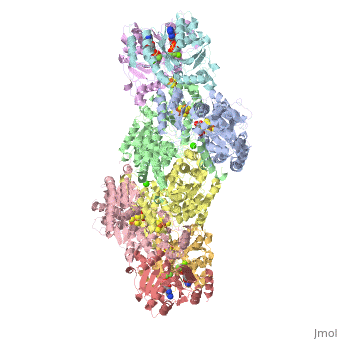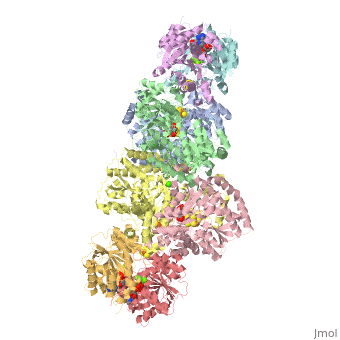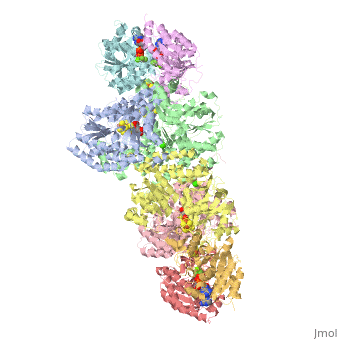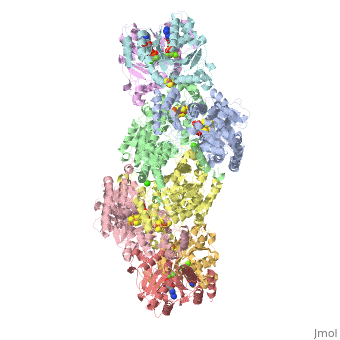
Nitrogenase
|
This is a default text for your page '. Click above on edit this page' to modify. Be careful with the < and > signs. You may include any references to papers as in: the use of JSmol in Proteopedia [1] or to the article describing Jmol [2] to the rescue.
Structural Highlights
The structure of nitrogenase consists of metal clusters centered throughout the protein. Three of these clusters are the 4Fe-4S Cluster, P-Cluster, and FeMo-Cluster. At either end of the protein are two copies of the Fe protein dimer, which is also the site of the ATP binding site. At these sites are ADP molecules which form a stable complex with the Fe protein. The MoFe protein, the central components of the protein, is where most of nitrogenase’s function is carried out. This protein requires a constant state of electrons which is supplied by the Fe protein which uses the hydrolysis of ATP to pump these electrons into the MoFe protein. Thus, it is helpful for the Fe protein to be coupled with ADP/ATP at the ends of the protein.
Fe-ATP Complex
| |||||||||||
References
Goodsell, David. “Nitrogenase.” PDB-101: Nitrogenase, Protein Data Bank, Feb. 2002, pdb101.rcsb.org/motm/26.