This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
User:Courtney Brown/Sandbox 1
From Proteopedia
The Human Histone H3/K9 Deacetylase, HDAC8
This is a default text for your page Courtney Brown/Sandbox 1. Click above on edit this page to modify. Be careful with the < and > signs. You may include any references to papers as in: the use of JSmol in Proteopedia [1] or to the article describing Jmol [2] to the rescue.
IntroductionHistonesHistones are a family of basic, positively charged proteins that associate with DNA inside the nucleus to help condense the DNA into chromatin [3]. The nuclear DNA is wrapped around the histone in order to fit in the nucleus. Nucleosomes are chromatin beads made up of DNA wrapped around eight histone proteins, or a histone octamer [3]. Modifying histones is a type of epigenetics, where changes are made in gene expression without altering the DNA sequence. Four different examples of modifying histones including histone acetylation, histone deacetylation, histone methylation and histone demethylation [3]. Histone Deacetylases (HDACs)HDACs are versatile enzymes that participate in regulating non-histone proteins, the cell cycle, cell differentiation and DNA-histone interactions, which are all steps that occur during the transformation of normal cell to malignant cancer cells. They preform ε-Amino-lysine acetylation, a type of histone modification that controls the stability of proteins and biological function in eukaryotic cells [4]. Histone deacetylation is the reversal process for this acetylation modification. There are different classes of HDACs based on phylogenetic analysis: •Class I - HDACs 1-3 and 8, which are homologous to yeast Rpd3 •Class II - HDACs 4-7, 9 and 10, which are homologous to yeast Hda1 •Class III - Sirtuin deacetylases •Class IV - HDAC 11 [4]. HDACs 1-11 are metalloenzymes and require a zinc ion for deacetylation due to their similar reaction mechanisms [4]. HDAC8Histone Deacetylase 8 is an enzyme found in Homo sapiens. HDAC8 is 388 residues long and consists of eight-stranded parallel β-sheets surrounded by 11 α-helices [4]. HDAC8 is the only functional HDAC that is found to be a single polypeptide instead of being high-molecular-weight multi-protein complexes [4]. HDAC8 decreases gene expression by increasing DNA-histone interactions, condensing the DNA tighter. StructureThe LigandThe bound to the HDAC8 includes an acetyl group, one arginine, one histidine, two lysines and MCM (a Coumarin fluorescence tag). It was derived from the p53 tumor suppressor gene. The substrate is held in the binding pocket via four interactions: three hydrogen bonds made with Asp101, and interactions with Gly151/Phe152/Phe208.
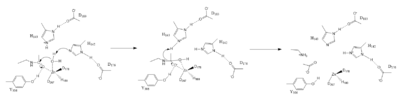
InhibitorPotassium Binding SiteActive SiteThe HDAC8 active site contains features of zinc and serine proteases. Substrates are deacetylated via a classic 2+ metal ion mechanism. Zinc pentacoordinates with , the nucleophilic water and the carbonyl oxygen of the substrate[4]. The Zinc ion by withdrawing electron density from the water due to the positive charge, making the water more acidic, therefore a better nucleophile. The Zinc ion also coordinates to the carbonyl oxygen of the acetyl group on the lysine, polarizing the carbonyl carbon, making it more electrophilic. There are two present in the active site. His142 deprotinates the water, the first step of the deacetylation (Figure 1). His 142 (stabilized by Asp 176) is closer to the water molecule than His 143 (stabilized by Asp 183), which was also thought to deprotinate the water. However, a mutation done to His143 only reduced activity, not abolished it, showing it is important but not crucial. His143 is instead thought to orient the substrate[4]. The attack by the deprotinated water forces the carbonyl carbon into a tetrehedral transition state. acts as the oxyanion hole, stabilizing this transition state via its protruding -OH group hydrogen-bonding with the negatively charged oxygen. The transition state collapses, and His 143 simultaneously gets deprotinated by the amide of the leaving acetyl group in order to weaken the scissle bond. Breaking this scissle bond results in a neutral lysine and a carboxylic acid, among other products. Body pH is around 7 typically and the pKa of carboxylic acids is around 2; the hydrogen from the COOH group is lost and picked up by the lysine, providing the hydrogen necessary for lysine to become acidic, per its nature[4]. DiseaseReferences
Student Contributors
| ||||||||||||