This is a default text for your page '. Click above on edit this page' to modify. Be careful with the < and > signs.
You may include any references to papers as in: the use of JSmol in Proteopedia [1] or to the article describing Jmol [2] to the rescue.
Function(s) and Biological Relevance
Lignostilbene-α,β-dioxygenase (LsdA) from the bacterium 'Sphingomonas paucimobilis'. It is a nonheme iron oxygenase that catalyzes the cleavage of lignostilbene, a compound
arising in lignin transformation, to two vanillin molecules. The substrate for this enzyme is lignostilbene. Phenylazophenol inhibited the LsdA-catalyzed cleavage of lignostilbene in a reversible, mixed fashion. Lignin is used in biofuel production. Lignin is a heterogeneous aromatic polymer found in plant cell walls that contributes to the recalcitrance of biomass. Below are two images. On the left is Lignostilbene, and on the right is Vanillin for comparison.
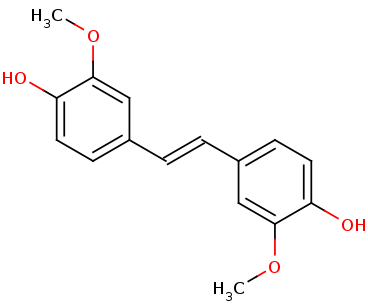
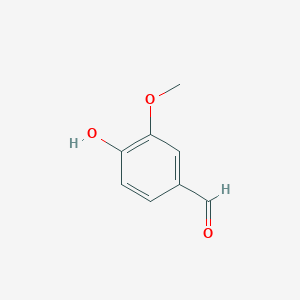
Broader Implications
To examine LsdA’s substrate specificity, we heterologously produce the dimeric enzyme with the help of chaperones. When tested on several substituted stilbenes, LsdA exhibited the greatest specificity for lignostilbene. These experiments further indicate that the substrate’s
4-hydroxy moiety is required for catalysis and that this moiety
cannot be replaced with a methoxy group. This expands our
mechanistic understanding of LsdA and related stilbene-cleaving dioxygenases.
Structural highlights and structure-function relationships
The of this protein is primarily made of the amino acids that are the main factor in catalysis. The 3 amino acids are Phe-59, Tyr101, and Lys-134. The is a fold of LsdA of a seven-bladed -propeller, typical of the carotenoid cleavage oxygenates (CCO's), which usually catalyze the oxidative cleavage of a double bond in carotenoids. The structures also consist of α-helices and ß-sheets. The occurs at the center of the propeller and contains an Fe2+. The and view of the ligand in the protein, which shows that both hydrophilic and hydrophobic residues are important to the ligand in the binding site.The space fill view allows us to see the different binding sites for this protein, the binding sites are allosteric. The for this protein is called NSL.
Energy Transformation
Phenylazophenol inhibits the LsdA-catalyzed cleavage of lignostilbene in a reversible, mixed fashion.


