Introduction
Vitamin K epoxide reductase (VKOR) is the enzyme responsible for regenerating vitamin K from vitamin K epoxide to support blood coagulation.
Vitamin K Cycle
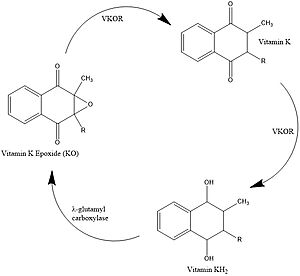
Figure 1. Vitamin K Cycle
Vitamin K is essential for blood clotting in the body. The fully reduced form, KH2, allows the gamma carboxylation of blood clotting cofactors and is turned into the epoxide form in the process. Vitamin K epoxide reductase, abbreviated VKOR, turns the epoxide back to the fully reduced form so the reduced form can be used again. This transformation happens in two steps including converting the epoxide to the partially oxidized Vitamin K quinone then converting the quinone to the fully reduced hydroquinone (KH2). [1]
Structural Overview
VKOR consists of four embedded in the endoplasmic reticulum membrane. The files on the RCSB Protein Data Bank include a barrel domain that is not pertinent to the function of VKOR. The images presented here have been edited to remove the barrel domain and renumbered to correspond with the article by Liu.
[2]. The original image with the barrel domain in context is shown in Figure 2.
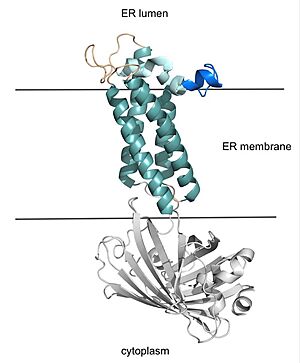
Figure 2. VKOR with Barrel Domain
Helices one and two are connected by the beta hairpin region which contains two of the active cysteines, C43 and C51. VKOR also has a cap domain covering the active site, made up of an anchor, loop, and helix.
Catalytic Cycle
Overview
The catalytic cycle of
Catalytic Cysteines
VKOR uses four catalytic cysteines (43, 51, 132, and 135) to facilitate reduction and cause conformational changes via disulfide bridge formation. When an oxidized or partially oxidized Vitamin K enters the active site, VKOR has a stabilizing 51-132 disulfide bond. C43 attacks that bond, forming a new bond with C51.
There are 4 catalytic cysteines that are important to VKOR, 43, 51, 132, and 135. To explain how this works, it is easiest to start with the second state. In the second state, an oxidized or partially oxidized Vitamin K has entered the active site. The stabilizing 51-132 disulfide bond is shown. Then in the third state, 43 has attacked the disulfide bond and made its own bond with 51. You can see 132 has an oxygen. That is because the researchers made a mutation from S to O to force the reaction to stop at that step so the structure could be deduced. In the natural VKOR, that would be a sulfur. The next state, the open state, results from 132 forming a bridge with 135. This allows release of the reduced or partially reduced Vitamin K. All of this disulfide rearranging was working to reduce the Vitamin K, particularly in the 135 position. If we go back to State 2, when Vitamin K first binds, you can see that 135 is not tied up in a disulfide bond. It is available to help the Vitamin K bond. So, it makes sense that once 135 gets forced to bond, the now reduced Vitamin K is released. State 5 is interesting because the disulfide bonds are similar to the open state, but warfarin is actually bound. This represents the binding of warfarin to the fully oxidized VKOR at the end of its cycle. Going back to State 1, the researchers used a mutation at 43 to mimic VKOR’s partially oxidized state. Warfarin can also bind to this state and notice that the disulfide bonds are the same as State 2. Also it is worth pointing out how the disulfide bonds contribute to conformational changes and are affected by conformational changes, which affects their proximity to each other and the active site.
Catalytic Amino Acids
Hydrophobic Interactions
Medical Relevance
Warfarin
Warfarin is a structural mimic of Vitamin K that is used clinically as an anticoagulant.
Superwarfarins


