Introduction
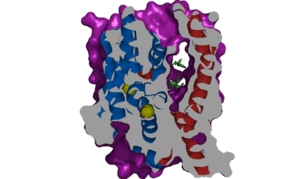
Structure
Active Sites
NTCP, among others in the SLC10 family, have . Many polar and negatively charged residues are characteristic of these active sites. The high level of conservation among sodium binding placement and interacting residues suggests sodium binding is coupled to bile salt transport. Additional mutations in the X-motif near sodium binding sites have shown that bile salt transport function is lost also suggesting that sodium allows bile salt binding.
[1] It is understood that these sodium binding sites facilitate changes from open-pore to closed pore states of NTCP that allow for the binding or release of bile salts. Closed-pore state is favored in the absence of sodium ions, while open-pore state is favored in the presence of sodium ions. This also allows for sodium concentrations to regulate uptake of taurocholates. When intracellular sodium levels are higher, open-pore state is favored allowing for the diffusion of taurocholates. However, when extracellular sodium levels are high, closed-state is favored preventing diffusion of taurocholates. [1]
The is also characteristic of NTCP. The pore surface remains Hydrophobic, while lining of the open pore state is largely Polar. The pattern of Hydrophobic and Polar is believed to follow similar patterns within taurocholate to prevent leakage of other bulky, amphipathic substrates. [1] When observing the relevant it is shown that some residues form Van der waals interactions while others will form dipole-dipole or ionic interactions with bile salt substrates. The core domain appears to contribute most of the polar domains, while the panel domain contributes more hydrophobic residues.
Conformational Change
Bile Salt Transport
Medical Relevancy
This is a sample scene created with SAT to by Group, and another to make of the protein. You can make your own scenes on SAT starting from scratch or loading and editing one of these sample scenes.