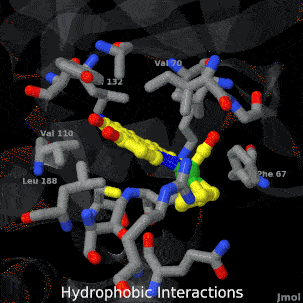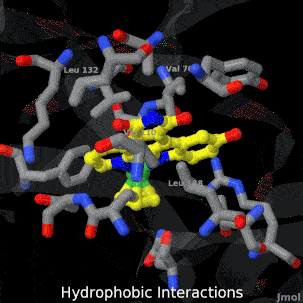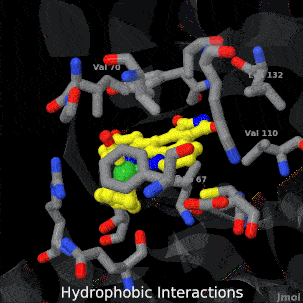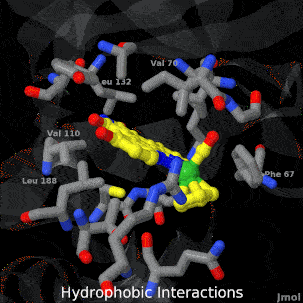
Sandbox Home
From Proteopedia
ISSN 2310-6301
As life is more than 2D, Proteopedia helps to bridge the gap between 3D structure & function of biomacromolecules
Proteopedia presents this information in a user-friendly way as a collaborative & free 3D-encyclopedia of proteins & other biomolecules.
|
||||||||
| Selected Research Pages | In Journals | Education | ||||||
|---|---|---|---|---|---|---|---|---|
|
|
|
||||||
|
||||||||