This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
1cma
From Proteopedia
|
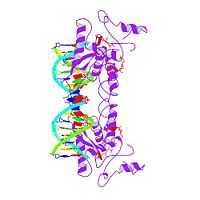
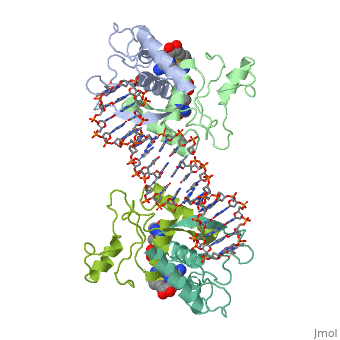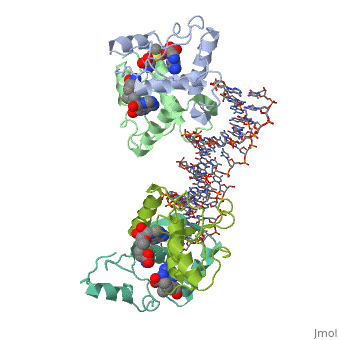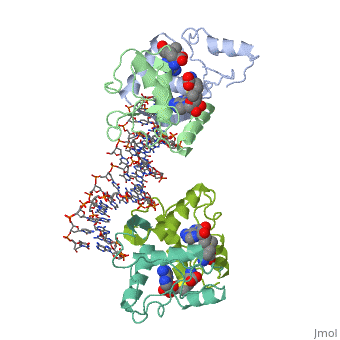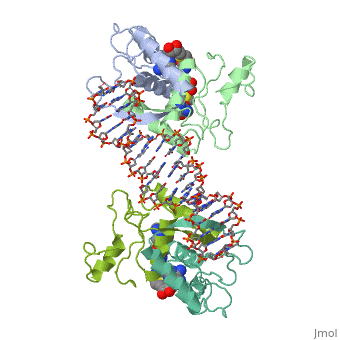
MET REPRESSOR/DNA COMPLEX + S-ADENOSYL-METHIONINE
Overview
The crystal structure of the met repressor-operator complex shows two, dimeric repressor molecules bound to adjacent sites 8 base pairs apart on, an 18-base-pair DNA fragment. Sequence specificity is achieved by, insertion of double-stranded antiparallel protein beta-ribbons into the, major groove of B-form DNA, with direct hydrogen-bonding between, amino-acid side chains and the base pairs. The repressor also recognizes, sequence-dependent distortion or flexibility of the operator phosphate, backbone, conferring specificity even for inaccessible base pairs.
About this Structure
1CMA is a Single protein structure of sequence from Escherichia coli with SAM as ligand. Full crystallographic information is available from OCA.
Reference
Crystal structure of the met repressor-operator complex at 2.8 A resolution reveals DNA recognition by beta-strands., Somers WS, Phillips SE, Nature. 1992 Oct 1;359(6394):387-93. PMID:1406951
Page seeded by OCA on Tue Nov 20 12:34:40 2007