This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
1ls8
From Proteopedia
|
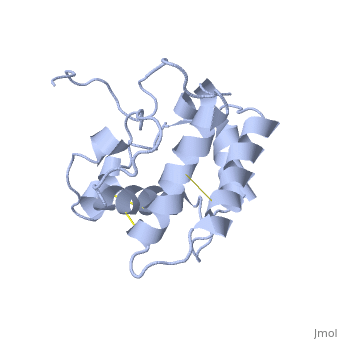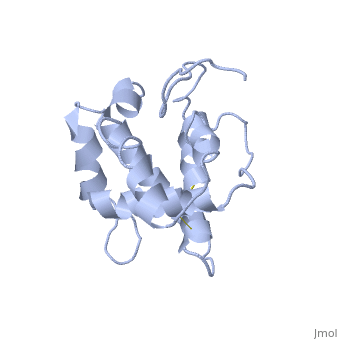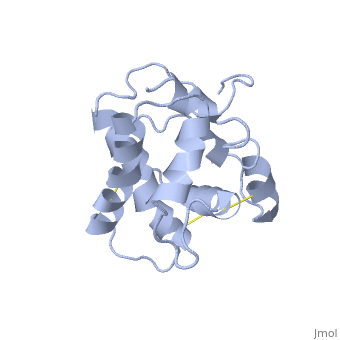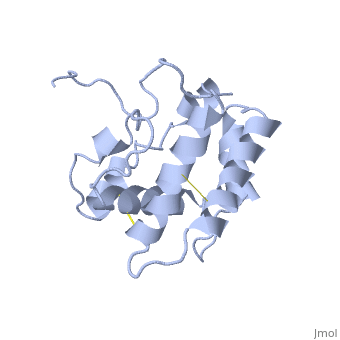
NMR structure of the unliganded Bombyx mori pheromone-binding protein at physiological pH
Overview
The nuclear magnetic resonance structure of the unliganded, pheromone-binding protein (PBP) from Bombyx mori at pH above 6.5, BmPBP(B), consists of seven helices with residues 3-8, 16-22, 29-32, 46-59, 70-79, 84-100, and 107-124, and contains the three disulfide, bridges 19-54, 50-108, and 97-117. This polypeptide fold encloses a large, hydrophobic cavity, with a sufficient volume to accommodate the natural, ligand bombykol. The polypeptide folds in free BmPBP(B) and in crystals of, a BmPBP-bombykol complex are nearly identical, indicating that the B-form, of BmPBP in solution represents the active conformation for ligand, binding.
About this Structure
1LS8 is a Single protein structure of sequence from Bombyx mori. Full crystallographic information is available from OCA.
Reference
NMR structure of the unliganded Bombyx mori pheromone-binding protein at physiological pH., Lee D, Damberger FF, Peng G, Horst R, Guntert P, Nikonova L, Leal WS, Wuthrich K, FEBS Lett. 2002 Nov 6;531(2):314-8. PMID:12417333
Page seeded by OCA on Tue Nov 20 20:49:51 2007