This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
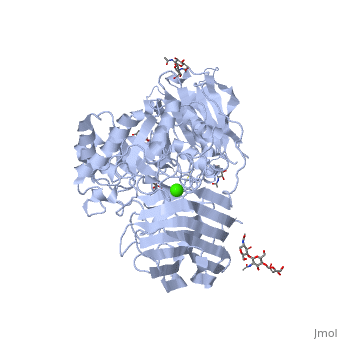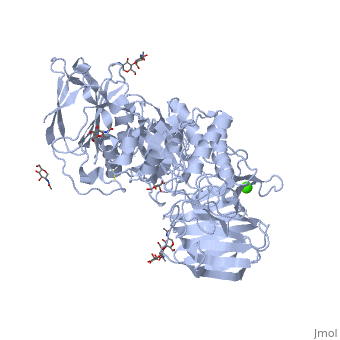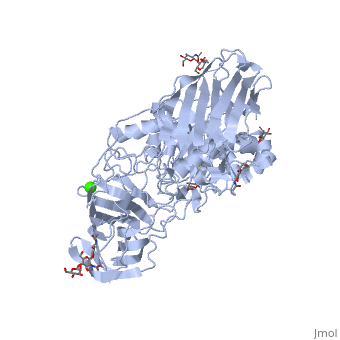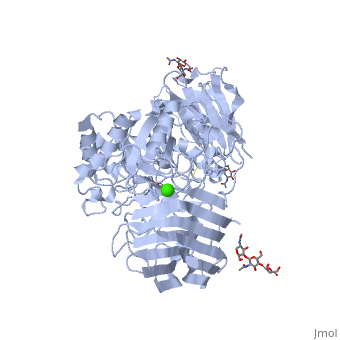
Galactosylceramidase
From Proteopedia
| |||||||||||
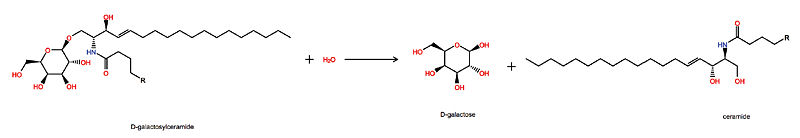
Krabbe disease is a devastating neurodegenerative disease characterized by widespread demyelination that is caused by defects in the enzyme galactocerebrosidase (GALC). Disease-causing mutations have been identified throughout the GALC gene. However, a molecular understanding of the effect of these mutations has been hampered by the lack of structural data for this enzyme. Here we present the crystal structures of GALC and the GALC-product complex, revealing a novel domain architecture with a previously uncharacterized lectin domain not observed in other hydrolases. All three domains of GALC contribute residues to the substrate-binding pocket, and disease-causing mutations are widely distributed throughout the protein. Our structures provide an essential insight into the diverse effects of pathogenic mutations on GALC function in human Krabbe variants and a compelling explanation for the severity of many mutations associated with fatal infantile disease. The localization of disease-associated mutations in the structure of GALC will facilitate identification of those patients that would be responsive to pharmacological chaperone therapies. Furthermore, our structure provides the atomic framework for the design of such drugs.
Insights into Krabbe disease from structures of galactocerebrosidase., Deane JE, Graham SC, Kim NN, Stein PE, McNair R, Cachon-Gonzalez MB, Cox TM, Read RJ, Proc Natl Acad Sci U S A. 2011 Sep 13;108(37):15169-73. Epub 2011 Aug 29. PMID:21876145
From MEDLINE®/PubMed®, a database of the U.S. National Library of Medicine.
Contents |
Ligands
- calcium ion
- N-acetyl-D-glucosamine
Inhibitors
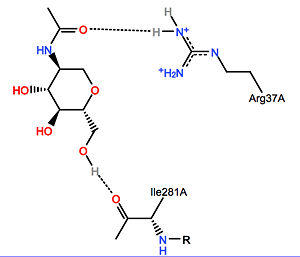
Because Krabbe disease is caused by a decrease in galactosylceramidase activity, there are no drug-protein interactions to explore. However some natural inhibitory molecules in humans include:
- 6-hexadecanoylamino-4-methylbelliferyl-beta-D-galactopyranoside, competitive inhibition
- D-galactose
- galactonyl hydrazide
- lactose
- N-(6-aminohexyl)-D-galactoside
- taurocholate (at high concentrations above 0.3% w/v)[8]
Quiz
3D Structures of galactosylceramidase
Updated on 15-January-2023
3zr5 – mGC – mouse
3zr6, 4cce – mGC + galactose
4ccc – mGC + nitrophenyl galactopyranose
4ccd – mGC + galactal
6y6s, 6y6t – mGC + galacto-noeurostegine
4ufh, 4ufi, 4ufj, 4ufl, 4ufk, 4ufm – mGC + azasugar inhibitor
5nxb – mGC + saposin A
References
- ↑ 1.0 1.1 RCSB Protein Data Bank - RCSB PDB - 3ZR5 Structure Summary. (n.d.). RCSB Protein Data Bank - RCSB PDB - 3ZR5 Structure Summary. Retrieved June 3, 2014, from www.rcsb.org DOI:10.2210/pdb3zr5/pdb
- ↑ Zizioli D, Guarienti M, Tobia C, Gariano G, Borsani G, Bresciani R, Ronca R, Giacopuzzi E, Preti A, Gaudenzi G, Belleri M, Di Salle E, Fabrias G, Casas J, Ribatti D, Monti E, Presta M. Molecular cloning and knockdown of galactocerebrosidase in zebrafish: new insights into the pathogenesis of Krabbe's disease. Biochim Biophys Acta. 2014 Apr;1842(4):665-75. doi: 10.1016/j.bbadis.2014.01.008., Epub 2014 Jan 24. PMID:24463171 doi:http://dx.doi.org/10.1016/j.bbadis.2014.01.008
- ↑ 3.0 3.1 3.2 3.3 3.4 Deane JE, Graham SC, Kim NN, Stein PE, McNair R, Cachon-Gonzalez MB, Cox TM, Read RJ. Insights into Krabbe disease from structures of galactocerebrosidase. Proc Natl Acad Sci U S A. 2011 Sep 13;108(37):15169-73. Epub 2011 Aug 29. PMID:21876145 doi:10.1073/pnas.1105639108
- ↑ Visigalli I, Ungari S, Martino S, Park H, Cesani M, Gentner B, Sergi Sergi L, Orlacchio A, Naldini L, Biffi A. The galactocerebrosidase enzyme contributes to the maintenance of a functional hematopoietic stem cell niche. Blood. 2010 Sep 16;116(11):1857-66. doi: 10.1182/blood-2009-12-256461. Epub 2010 , May 28. PMID:20511539 doi:http://dx.doi.org/10.1182/blood-2009-12-256461
- ↑ 5.0 5.1 Belleri M, Ronca R, Coltrini D, Nico B, Ribatti D, Poliani PL, Giacomini A, Alessi P, Marchesini S, Santos MB, Bongarzone ER, Presta M. Inhibition of angiogenesis by beta-galactosylceramidase deficiency in globoid cell leukodystrophy. Brain. 2013 Sep;136(Pt 9):2859-75. doi: 10.1093/brain/awt215. PMID:23983033 doi:http://dx.doi.org/10.1093/brain/awt215
- ↑ 6.0 6.1 6.2 Kohlschutter A. Lysosomal leukodystrophies: Krabbe disease and metachromatic leukodystrophy. Handb Clin Neurol. 2013;113:1611-8. doi: 10.1016/B978-0-444-59565-2.00029-0. PMID:23622382 doi:http://dx.doi.org/10.1016/B978-0-444-59565-2.00029-0
- ↑ 7.0 7.1 7.2 Teixeira CA, Miranda CO, Sousa VF, Santos TE, Malheiro AR, Solomon M, Maegawa GH, Brites P, Sousa MM. Early axonal loss accompanied by impaired endocytosis, abnormal axonal transport, and decreased microtubule stability occur in the model of Krabbe's disease. Neurobiol Dis. 2014 Jun;66:92-103. doi: 10.1016/j.nbd.2014.02.012. Epub 2014 Mar , 6. PMID:24607884 doi:http://dx.doi.org/10.1016/j.nbd.2014.02.012
- ↑ EC 3.2.1.46 - galactosylceramidase. (n.d.). Information on. Retrieved June 3, 2014, from www.brenda-enzymes.org
Proteopedia Page Contributors and Editors (what is this?)
Alison Stivers, Michal Harel, Dillon Shapiro, Angel Herraez, Joel L. Sussman