This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
NF-Y Transcription Factor Sandbox
From Proteopedia
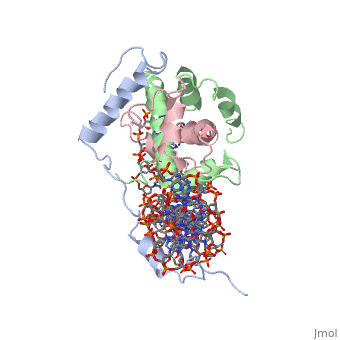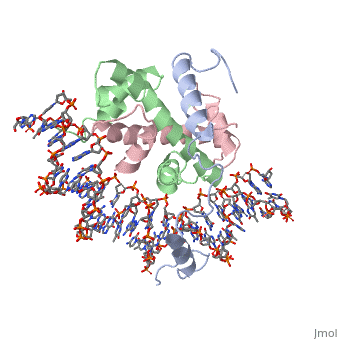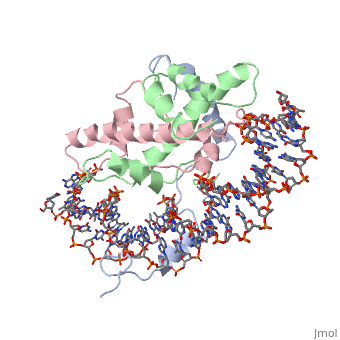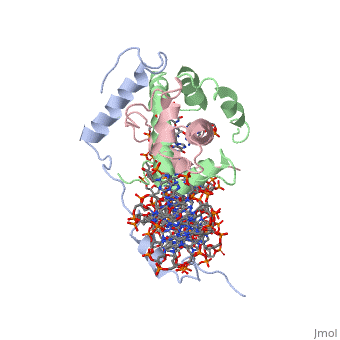
OverviewA transcription factor (TF) is a protein that binds to specific DNA sequences and can either repress or activate the transcription of a gene. TFs have a diverse family of proteins and normally exist in a multisubunit complex. NF-Y is a transcription factor involved in histone post-translational modifications (PTMs) [1]. In plants, the NF-Y transcription factors regulate and respond to many physiological responses. NF-Y subunits are closely related to core histones. A histone is a conserved protein that wraps 146 nucleotides of DNA into the basic unit of chromatin, the nucleosome [2]. Histone-fold Domains (HFDs) are required for the tertiary structure of histones and non-sequence specific contacts with DNA[2]. Protein StructureThe NF-Y transcription factor consists of , , and subunits. NF-YA subunit contains two α-helices, NF-YB subunit contains four α-helices and two β-sheets, and NF-YC subunit contains three α-helices and two β-sheets. The NF-YB and NF-YC subunits each contain a histone fold motif and form a NF-YB/NF-YC histone folding domain (HFD) dimer[3]. The composition of mostly α-helices gives the protein flexibility. One of the two α helices of the NF-YA subunit, the N terminal , interacts with NF-YB/NF-YC heterodimer resulting in a heterotrimer.
Protein FunctionThe post-translational modifications (PTMs) that NF-Y transcription factor is associated with aid in identifying regions of DNA that are destined to be transcribed. NF-Y is responsible for recruiting enzymes responsible for transcription (like RNA Polymerase II), and enzymes involved in acetylations on active promoters, suggesting that NF-Y is involved in switch-modifications [2]. Furthermore, NF-Y is a sequence-specific TF. It is possible that NF-Y and other sequence-specific TFs determine histone modifications on promoters[1]. NF-Y is regulated by redox mechanisms[4]. The regulated subunit (NF-YB) has three conserved cysteines in its A2 helix: , , and ; which sense the cellular redox potential and allow heterodimerization under reduced conditions. In oxidized conditions, NF-YB forms heterodimers in the cytoplasm which hinders CCAAT-binding and transcriptional activation[4].
DNA InteractionNF-Y interacts with DNA in several ways; one particular way is by using the C terminal of the NF-YA subunit inserts deep into the minor groove of DNA. NF-YA A2 helix binds to the box and causes the minor groove to widen at the CCAAT box[1]. residues interacting with the CCAAT box prevent G bases due to steric reasons, and these residues perform specific interactions that link the NF-Y/DNA complex. Van der Waals and provide the stabilization of the NF-Y/DNA complex due to the highly basic surface of the NF-YB/NF-YC HFD dimer and negatively charged DNA[1](Hydrophobic Polar). Interaction between NF-Y and DNA can be blocked by drugs that bind to the minor groove. References
| ||||||||||||