This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Platelet glycoprotein
From Proteopedia
(Redirected from Platelet-receptor glycoprotein Ib alpha)
| |||||||||||
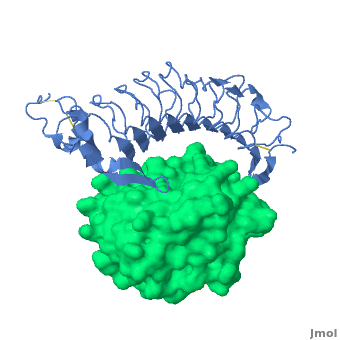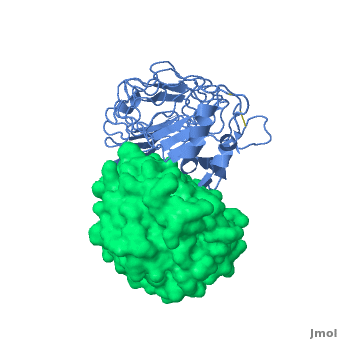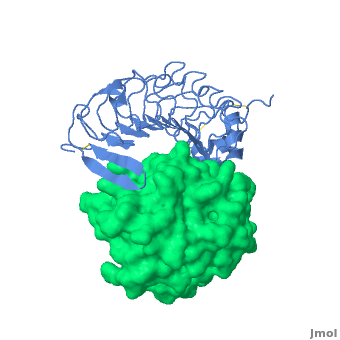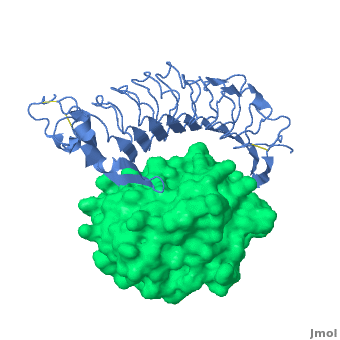
3D structures of platelet glycoprotein
Updated on 03-September-2023
Reference
- Huizinga EG, Tsuji S, Romijn RA, Schiphorst ME, de Groot PG, Sixma JJ, Gros P. Structures of glycoprotein Ibalpha and its complex with von Willebrand factor A1 domain. Science. 2002 Aug 16;297(5584):1176-9. PMID:12183630 doi:10.1126/science.107355
See Also
Proteopedia Page Contributors and Editors (what is this?)
Michal Harel, Alexander Berchansky, Joel L. Sussman, Wayne Decatur, Jaime Prilusky