This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
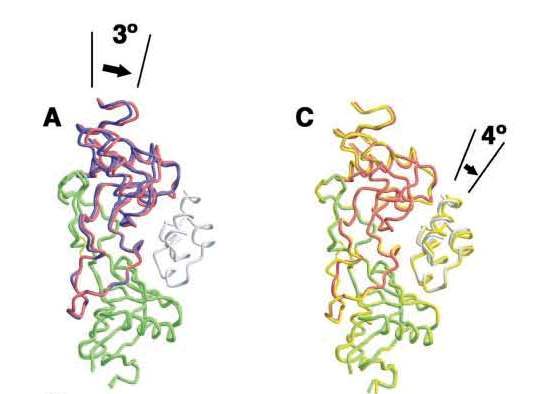
Rotation of 4 degrees of FIX
From Proteopedia
Movement of A chain of IX-bp caused by induced fitting and of IXGD1–46 caused by Mg2+ ions [1]. A, interaction patch for IX-bp (residues 100–110 of IX-bp B chain) is overlaid. Complexed and uncomplexed IX-bp A and B chains are shown in red and green for A and in blue and green for B, respectively. B, hydrophobic and hydrophilic interactions involving Lys22 of IXGD1–46 and Thr62, Lys63, and Glu108 of IX-bp A chain resulting from the movement of the A chain. The surface model was calculated using GRASP (41). This interaction was not observed, formed under Mg2+-free condition. C, Mg2+-bound IXGD1–46/IX-bp complex is shown in red and green, and the Mg2+-free (Ca2+ only) complex is depicted by yellow ribbon representation. Both structures are superimposed as a result of the position of C atoms of IX-bp.
Additional Resources
For additional information, see: Hemophilia
References
- ↑ Shikamoto Y, Morita T, Fujimoto Z, Mizuno H. Crystal structure of Mg2+- and Ca2+-bound Gla domain of factor IX complexed with binding protein. J Biol Chem. 2003 Jun 27;278(26):24090-4. Epub 2003 Apr 14. PMID:12695512 doi:10.1074/jbc.M300650200