| The SRY protein is a 204 residues-long monomeric polypeptide. It is encoded by the SRY gene and is involved in the sex determination in mammals by being responsible for the gonadogenesis and so the male sexual development. It is the HMG-box that gives to the protein its ability to bind DNA by its minor groove[1]. |
| Related: | 1hrz |
| Resources: | FirstGlance, OCA, PDBe, RCSB, PDBsum
|
| Encoded by | The SRY gene codes for the SRY protein
|
| Length | 204 residues
|
| Molecular weight | ≈23kDa
|
| DNA target sequence | (5'-dGCACAAAC)
|
| Regulation | Sf1;Sp1 (binding site in the promoter of SRY : -150),WT1 (binding site in the promoter of SRY : -87)
|
| Role | Transcription factor
|
| Genes targeted | SOX9, TH, MAOA
|
History
After centuries of unfounded theories mainly based on environmental factors, the first molecular theory concerning the sex determination appeared in 1891. At this time, the german biologist Hermann Henking was studying sperm formation in wasps. As a chromosome which was not present in all the wasps looked different from the others, he suspected it to play a role in sex determination and called it the "X chromosome".
Ten years later, Clarence Erwin McClung saw that this chromosome behaved differently during the meiosis and was only present in half the sperm cells of grasshoppers. As the main characteristic that varies in 50/50 proportions among zygotes is the sex, McClung suspected the X chromosome to be implicated in sexual development.
In 1905, Nettie Stevens discovered the "Y chromosome" (and the female XX and male XY patterns) while she was counting the chromosomes of beetles under the microscope[2].
During the next decades, a few theories were in competition. In 1921, Calvin Bridges's works on Drosophila melanogaster seemed to reveal that male characters acquisition is due to a genic balance between the genes contained in the X chromosome and those contained in the autosomes[3].
In 1930, Ronald Fisher introduced the first Y-based control of sex theory by proposing two different models : either all the genes responsible for the male characters are located on the Y chromosome or there is a Y-located gene which regulates the expression of genes elsewhere in the genome[4].
As Alfred Jost had shown the testosterone produced by the testis is responsible for the entire male phenotype acquisition[5], Peter Neville Goodfellow proposed in 1988, that there is a gene (TDF in human, Tdy in mice) on the Y chromosome which drives the development of the testis[4]. In 1990, Goodfellow's hypothesis was validated with the discovery of Tdy's localisation. This gene's product (expressed during the male gonadal development) owns an amino-acid motif which shows homology to other known or putative DNA-binding domains. Tdy is therefore a transcriptional factor[6]. The same year, the human SRY gene (accepted later as the TDF) was discovered[7].
Three dimensional structure of the SRY protein was determined in 1995 using NMR spectroscopy[8].
SRY gene
The SRY gene encodes the SRY protein. The SRY protein is a transcriptional factor that induces the male phenotype in the embryo. The SRY gene is located on the Y chromosome in the short arm (p) 11.3 [9]. This gene has only one exon that contains the HMG domain (DNA-binding High-Mobility Group box domain). It means that SRY mRNA does not have an alternative splicing, so there is only one isoforme of SRY protein[10]. Moreover, the human genome contains only one copy of the SRY gene, whereas the mouse genome contains 6 copy of this gene[11].
Sequence of the SRY gene
>gi|568815574:c2787741-2786855 Homo sapiens chromosome Y, GRCh38.p2 Primary Assembly [12]
TGTTGAGGGCGGAGAAATGCAAGTTTCATTACAAAAGTTAACGTAACAAAGAATCTGGTAGAAGTGAGTT
TTGGATAGTAAAATAAGTTTCGAACTCTGGCACCTTTCAATTTTGTCGCACTCTCCTTGTTTTTGACA
ATGCAATCATATGCTTCTGCTATGTTAAGCGTATTCAACAGCGATGATTACAGTCCAGCTGTGCAAGAGAAT
ATTCCCGCTCTCCGGAGAAGCTCTTCCTTCCTTTGCACTGAAAGCTGTAACTCTAAGTATCAGTGTGAAA
CGGGAGAAAACAGTAAAGGCAACGTCCAGGATAGAGTGAAGCGACCCATGAACGCATTCATCGTGTGGTC
TCGCGATCAGAGGCGCAAGATGGCTCTAGAGAATCCCAGAATGCGAAACTCAGAGATCAGCAAGCAGCTG
GGATACCAGTGGAAAATGCTTACTGAAGCCGAAAAATGGCCATTCTTCCAGGAGGCACAGAAATTACAGG
CCATGCACAGAGAGAAATACCCGAATTATAAGTATCGACCTCGTCGGAAGGCGAAGATGCTGCCGAAGAA
TTGCAGTTTGCTTCCCGCAGATCCCGCTTCGGTACTCTGCAGCGAAGTGCAACTGGACAACAGGTTGTAC
AGGGATGACTGTACGAAAGCCACACACTCAAGAATGGAGCACCAGCTAGGCCACTTACCGCCCATCAACG
CAGCCAGCTCACCGCAGCAACGGGACCGCTACAGCCACTGGACAAAGCTGTAGGACAATCGGGTAACATT
GGCTACAAAGACCTACCTAGATGCTCCTTTTTACGATAACTTACAGCCCTCACTTTCTTATGTTTAGTTT
CAATATTGTTTTCTTTTCTCTGGCTAATAAAGGCCTTATTCATTTCA
(Legend : Initiation codon ; HMG sequence ; Stop codon)
Regulation of the expression of the SRY gene
In humans, the SRY promoter is located between −408 and −95 bp. Moreover, the SRY gene has enhancers at -727 pb. The linkage between regulatory proteins and these enhancers has the property to increase the production of SRY proteins. These regulatory proteins could be: SF1 (steroidogenic factor 1), SP1 and WT 1 (Wilms tumor)[13].
- SF1: this transcriptional factor belongs to the family of nuclear hormone receptors and contains a zinc finger. The activation of this protein requires a ligand (hormone).
- SP1: this transcriptional factor is an ubiquitous protein which binds rich GC-sites and is implicated in the transcription of many genes. Moreover, this protein contains a zinc finger.
- WT1: this transcriptional factor transactivates the SRY gene. It contains a zinc finger[14].
Structure
The SRY-HMG domain (HMG-Box)
SRY-HMG stands for Sex determining Region Y - High Mobility Group domain.
It is approximately 80 residues-long. It mediates the binding of the protein to the minor groove of DNA. It is the most important part of the SRY protein. Not only because it enables the protein to bind the DNA but also because even a single mutation can cause an inactivation of the protein.
It has a twisted L shape: it has a long (28Å) and a short (22Å) arm. The HMG Box is made of 3 helices. Its N-term and C-term domains are irregular. The overall structure is stabilized by a hydrophobic core especially at the intersection of the 3 helices where 3 aromatics cycles meet, surrounded by aliphatic aminoacids.
See the different structures:
The interaction between the HMG-Box and DNA is specific and stable. It is mostly hydrophobic. It permits the DNA to bend of DNA (≈75°). Only one molecule of water interfaces the Box and the DNA. The complex is stabilized by salt bridges between positively charged residues of the HMG domain and negatively charged phosphates[1].
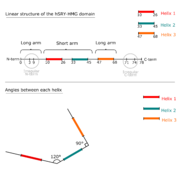
Linear structure of hHMG domain
The binding of SRY on DNA is specific. The DNA target site is a DNA octamer :
(5'-dGCACAAAC)
(5'-dGTTTGTGC)
This sequence is found in the promoters of genes expressed during the testicular development.
The bend of DNA enables the recruitment of different proteins and the building of massive proteins-DNA complexes that could change the expression of different genes[15].
Even if the most important function of the HMG box is binding and bending DNA, it is also involved in DNA condensation, recombination and repair.
There are two kinds of proteins that contain a HMG box:
- HMG1 : It is expressed in few cell types. It is found in transcription factors that contain a single HMG box. The binding of a DNA sequence is specific.
- HMG2 : It is found in all cell types and is abundant in chromatin. These proteins can contain two or more HMG boxes that can non-specifically bind DNA.
General structure of SRY
The overall structure of SRY is organized around the HMG-box.
3 domains:
- N-term domain
- Central domain : DNA binding (HMG box)
- C-term domain
Function
Sex determining
It acts like a sex determining factor thanks to its transcriptional activity. It inhibits the development of female sex structures in the embryonic individual.
The SRY protein contains nuclear localization signals in N and C terminals. An acetylation of these domains allows the exportation of the SRY protein to the nucleus[13].
The SRY protein activates the SOX9 (SRY-box9) gene [10]. This gene is found in long arm 24.3 of the chromosome 17[16] and is implicated in the stimulation of the differentiation of pre-Sertori into Sertoli cells rather than granulosa cells.
The activation of SOX9 is done by the SRY protein and another transcriptional factor: SF1 (steroidogenic factor 1). These transcriptional factors are bound on an enhancer called: TESCO (Testis-Specific Enhancer of SOX9 core element). The binding of a transcriptional factor on an enhancer provokes a curvature of the DNA (≈75°), allowing a stabilization of the elongation complex on the SOX9 promoter. The SOX9 protein activates the gene AMH (Anti-Mullerian Hormone)[17]. Therefore, it allows the degeneration of the channels of Müller in male[13].
Cathecolamines regulation
It has been shown that SRY is present in brain regions and activates the Tyrosine-3-Hydroxylase expression. This enzyme catalyses the rate-limiting step of catecholamine synthesis (L-Tyrosine to L-DOPA). Furthermore, SRY also activates the expression of Monoamine Oxidase A which is responsible for the inactivation of catecholamines. Therefore, it regulates both positively and negatively the catecholamines concentration[18].
Other extra-testicular effects
Some knock-down experiments have shown that SRY may also be a major actor in the dopamine pathway. On the peripheral side, it seems to regulate noradrenaline levels and blood pressure[18]. A researcher who contributed to discovering these effects says it might explain why "the aggressive fight-or-flight reaction is more dominant in men, while women predominantly adopt a less aggressive tend-and-befriend response"[19].
It has been shown that SRY may be involved in the regulation of the renin-angiotensin system in mice. Indeed, sequence mutations of the protein lead to hypertension. Because the human's SRY organisation is very closed to the mouse's, it has been proposed that SRY may have the same function in humans but it has not been studied directly[20].
Diseases
Swyer syndrome (AKA XY gonadal dysgenis)
If the TDF protein is not able to bind its targeted DNA sequences, the genes responsible for the testis development are not expressed. The patient owning this defective protein will then develop female characters, even though he has a XY karyotype. This phenomenon is known as the "Swyer Syndrome".
Different causes can explain this "XY gonadal dysgenis", as it is also called. About thirty mutations (named "SRXY1") in the SRY gene have been shown to drive this phenotype development. It can also be due to crossovers during a meiosis. If a Y chromosome portion carrying the SRY gene is recombined into a X chromosome, a sperm cell will get this abnormal Y chromosome. If it then fecundates, a XY karyotype without any SRY gene will be formed.
De La Chapelle syndrome (AKA XX male syndrome)
From the meiosis just described would also result an abnormal X chromosome, carrying the SRY gene. If the sperm cell owning this chromosome fecundates an ovule, the resulting newborn will have a XX karyotype but a male phenotype. This is called the "De La Chapelle syndrome". In this case, the patient can either develop testis or both testis and ovarian tissues. As some epigenetic mechanisms can inactivate the X chromosome carrying SRY, this syndrome keeps most of the patients sterile[21].
Hepatocellular carcinoma
In the case on a hepatocellular carcinoma, it has been proved that high expression levels of SRY help cancer progression and poor patient survival. However, it still seems that hepatocellular carcinoma is not most likely to develop in men[22].
Structural highlights
This is a sample scene created with SAT to by Group, and another to make of the protein. You can make your own scenes on SAT starting from scratch or loading and editing one of these sample scenes.

