Introduction
Lysophosphatidic Acid Receptor 1 (commonly referred to as LPA1) is a G protein-coupled receptor and one of 6 different LPA receptors (LPA1-LPA6). These receptors bind the phospholipid derivative lysophosphatidic acid (LPA), a signaling molecule that acts as a potent mitogen upon binding to one of its six receptors.[1] LPA1 is part of the larger EDG receptor family, which includes the more widely studied sphingosine 1-phopshate receptors.[1] This receptor is responsible for initiating several different signaling cascades with different molecules and G-proteins.[2] These cascades ultimately result in growth, survival, and movement of cells, as well as neural cell development.[3]
Structure
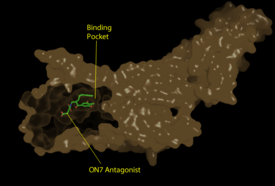
Figure 1: Surface representation of the LPA
1 receptor in tan interacting with its antagonist, ON7, shown in green and red sticks.The exterior of the protein was partially cut away to display the interior binding pocket.
The LPA1 receptor protein is composed of 364 amino acids with a molecular weight of approximately 41 kDa. Common to all G-protein coupled receptors, LPA1 contains seven alpha helices which make up the seven transmembrane spanning domains with three intracellular loops and three extracellular loops.[4] Within these helices is an interior binding pocket that stabilizes the binding of LPA1's natural ligand, LPA (Figure 1). The opening to this binding pocket is larger than other LPA receptors, enabling this receptor to bind ligands other than its natural ligand, such as 2-AG.[1]
Key Ligand Interactions
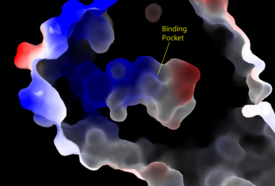
Figure 2: Electrostatic illustration of the amphipathic binding pocket of the LPA
1 receptor. This binding pocket was revealed by cutting away the exterior or the protein. This binding pocket, located in the interior of the protein, has both polar and nonpolar regions. The blue and red coloration highlight the positively and negatively charged regions, respectively, and the white color shows the nonpolar region of the binding pocket.
The biological ligand of the LPA1 receptor receptor is lysophosphatidic acid (LPA), a phospholipid that contains a long, nonpolar tail, a phosphate head, a chiral hydroxyl group, and an ester group. This receptor provides specificity for its ligand by the amphipathic binding pocket; the positive region on the left hand side of the pocket stabilizes the LPA's phosphate group, the nonpolar region at the bottom of the binding pocket stabilizes the hydrophobic tail of LPA, and the polar region at the top of the pocket stabilize binding of the ester and hydroxyl group (Figure 2). ONO-9780307 (ON7) is an antagonist for LPA due to its large nonpolar region, chiral hydroxyl group, ester, and carboxylic acid which all resemble portions of the LPA molecule (Figure 3). Four separate interactions with this antagonist of LPA1 help demonstrate the key interactions that stabilize the binding of the LPA phospholipid to this receptor. In the nonpolar region of the binding pocket, of LPA1 stabilize the large nonpolar group of ON7. At the polar region, the ligand binding is stabilized by forming ionic and polar interactions with the carboxylic acid and the hydroxyl group of ON7.[1] In addition, interplay between causes another stabilizing component with the ON7 antagonist. Glu293 forms polar interactions with Lys39, positioning it in close proximity to to the carboxylic acid of ON7, which then interactions with Lys39 via ionic bonding.[1] While Lys39 is highly conserved among all six LPA receptors, a neighboring histidine residue is specific to the LPA1 receptor. forms both ionic and polar interactions with the carboxylic acid of ON7. Protonation of this residue greatly affects the binding affinity of LPA, leading to an increase in the pathways associated with cell proliferation and migration. Because cancerous tumors create acidic environments where His40 is protonated, this residue is an important link to tumor growth and cancer cell movement.[1]
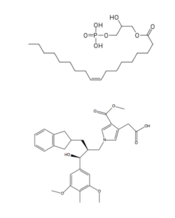
Figure 3: Structures of LPA and its antagonist, ON7, respectively.
Function
The LPA1 receptor is present in nearly all cells and tissues, and deletion of the LPA1 receptor has physiological effects on every organ system, indicating its wide range of functions. Specifically, this receptor initiates downstream signaling cascades with three Gα proteins: Gi/o, Gq/11, and G12/13. Specifically, Gα proteins begin signaling cascades that activate phospholipase C and MAPKs that signal for cell proliferation, survival, and migration. [2] Despite this receptor being expressed ubiquitously, LPA1 is highly expressed in neural tissue, aiding in several different neurodevelopmental processes including growth and folding of the cerebral cortex and the growth, survival, and migration of neural progenitor cells.[3] Finally, LPA1 receptors expressed in neural tissue act through a signaling pathway with the Rac1 G-protein to aid in Schwann cell migration and myelination.[5]
Receptor Comparison
Sphingosine 1-Phosphate Receptor
LPA1 belongs to the EDG (endothelial differentiation gene) family of lysophospholipid receptors. This family also includes the sphingosine 1-phosphate receptor 1 (S1P1), which shares many structural similarities to LPA1. In fact, the transmembrane regions share a sequence identity of 41%. [6] A defining difference between these two receptors is their mode of ligand access to the binding site. The hydrophobic S1P ligand enters S1P1 via the membrane. LPA1 differs and utilizes an extracellular opening that allows LPA access from the extracellular space (Figure 4). [1] Structural evidence for this altered ligand binding pathway includes global changes in the positioning of the extracellular loops (ECL) and transmembrane helices (TM). Specifically, a slight divergence of , which is positioned 3 Å closer to TMVII compared to S1P1, and a repositioning of , resulting in a divergence of 8 Å from S1P1 result in ligand access via the extracellular space. [1] This narrowing of the gap between TMI and TMVII blocks membrane ligand access in LPA1, while the greater distance between ECL3 and the other extracellular loops promotes extracellular access for LPA1. Additionally, ECL0 is helical in S1P1, but in LPA1. This increased flexibility that results from ECL0 lack of secondary structure in LPA1 further promotes favorable LPA access to the binding pocket from the extracellular space. [1]
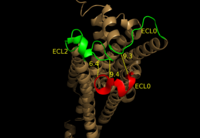
Figure 4: Extracellular ligand access pathway on LPA
1. Compared to S1P
1, ECL3 shown in red is positioned 8 Å further from ECL0 and ECL2 shown in green. This largely contributes to the altered ligand binding pathway between LPA
1, and S1P
1 Endocannabinoid Receptor 1
LPA1 is also closely related to the first of the six cannabinoid receptors. This close relation gives CB1 (Cannabinoid Receptor 1) the ability to bind to analogs of LPA and LPA1 the ability to bind to analogs of CB1 ligands. [1] This crossing over of ligand binding opens the possibility of metabolic crosstalk between the two signaling systems. [1] Complementary access to the LPA1 binding pocket can be achieved by phosphorylated CB1 ligand analogs, while complementary access to the CB1 binding site requires dephosphorylation of LPA1 ligand analogs (Figure 5). In both cases, a ligand could serve as a primary receptor modulator and a simultaneous prodrug for a different receptor. [1]
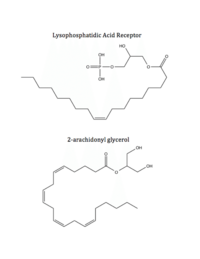
Figure 5: Respective ligands of LPA
1 and CB
1. LPA binds to CB
1 after dephosphorylation, while 2-AG binds to LPA
1 after phosphorylation.
located within the hydrophobic binding pocket of LPA1 may share responsibility for the preference for long unsaturated acyl chains, including the ligand LPA. The polarity of these residues provide favorable interactions between the ligand and the binding pocket.[1] Additionally, Asp129 and Trp210 may serve as a trigger for agonist induced conformational changes. These residues are also interesting in regard to GPCR phylogenic evolution. [1] Trp210 specifically only occurs in this position in 1% of all class A GPCR receptors and is unique to lysophospholipid and cannabinoid receptors. [7] A model for lipid agonist binding generated through molecular modeling was used to dock two of the cannabinoid receptor CB1's most abundant endogenous ligands into the LPA1 binding pocket. [1] Rotameric shifts of Trp210 and Trp271 lead to the expansion of the binding pocket and the exposure of the π clouds of their indole rings. These shifts and expansion provided favorable interactions with the double bonds of the phosphorylated cannabinoid ligands (Figure 6). This favorable binding provided evidence that the hydrophobic binding pockets of LPA1 and CB1 are able to favorably bind the same poly-unsaturated acyl chains with metabolically interconvertible head groups. [1]
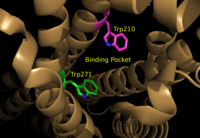
Figure 6: Illustration of key LPA
1 binding pocket residues Trp210 and Trp271. These residues provide rotameric shifts and expansion that allow for favorable interactions with phosphorylated cannabinoid ligands.
Disease Relevance
Because LPA1 is expressed in so many tissues throughout the body, LPA1 has been linked to the symptoms and progression of several different diseases and disorders.[8][9] For example, due to LPA1's role in pain signaling, overexpression of this protein can cause both allodynia or hyperalgesia, common symptoms of multiple sclerosis or strokes.[8] In addition, since LPA1 helps in the myelination of Schwann cells, mutation of the receptor can also lead to a decrease in prepulse inhibition, a general sign of schizophrenia.[8] Lastly, because of the mitogen signaling activity of LPA1, abnormal expression or mutation of this receptor has been linked to tumor growth, survival, and migration in both liver and lung tumors.[9] Part of this tumorigenesis can be explained by the action of on LPA1 where, when protonated, increases LPA binding affinity by up to 1kcal/mol.[1] Consequently, in the acidic environment produced by hypoxic tumors creating lactic acid, LPA1 activity is increased, allowing these tumors to continue to proliferate, migrate, and survive.[9] In contrast, the LPA1 receptor also induces protective functions in different cardiovascular conditions. For example, in patients with heart disease, LPA1 communicates with PI3K, PKB, and ERK to create a hypertrophic response in the heart to offset reduced heart contractions.[8]






