This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Sandbox Reserved 1647
From Proteopedia
| |||||||||||
References
- ↑ TBX21/T-Bet polyclonal antibody : https://www.thermofisher.com/antibody/product/TBX21-T-bet-Antibody-Polyclonal/13700-1-AP
- ↑ TBX21-GeneCards : TBX21 - T-box Transcription factor 21 :https://www.genecards.org/cgi-bin/carddisp.pl?gene=TBX21
- ↑ Mehta DS, Wurster AL, Weinmann AS, Grusby MJ. NFATc2 and T-bet contribute to T-helper-cell-subset-specific regulation of IL-21 expression. Proc Natl Acad Sci U S A. 2005 Feb 8;102(6):2016-21. doi:, 10.1073/pnas.0409512102. Epub 2005 Jan 31. PMID:15684054 doi:http://dx.doi.org/10.1073/pnas.0409512102
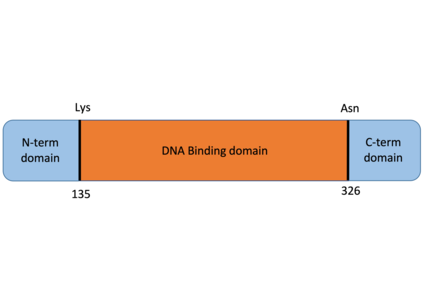
- ↑ Jang EJ, Park HR, Hong JH, Hwang ES. Lysine 313 of T-box is crucial for modulation of protein stability, DNA binding, and threonine phosphorylation of T-bet. J Immunol. 2013 Jun 1;190(11):5764-70. doi: 10.4049/jimmunol.1203403. Epub 2013, Apr 24. PMID:23616576 doi:http://dx.doi.org/10.4049/jimmunol.1203403
- ↑ Wang P, Wang Y, Xie L, Xiao M, Wu J, Xu L, Bai Q, Hao Y, Huang Q, Chen X, He R, Li B, Yang S, Chen Y, Wu Y, Ye L. The Transcription Factor T-Bet Is Required for Optimal Type I Follicular Helper T Cell Maintenance During Acute Viral Infection. Front Immunol. 2019 Mar 29;10:606. doi: 10.3389/fimmu.2019.00606. eCollection, 2019. PMID:30984183 doi:http://dx.doi.org/10.3389/fimmu.2019.00606
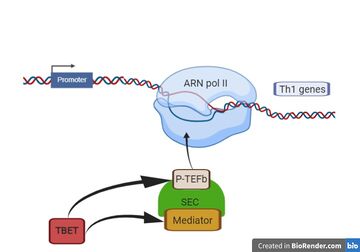
- ↑ Hertweck A, Evans CM, Eskandarpour M, Lau JC, Oleinika K, Jackson I, Kelly A, Ambrose J, Adamson P, Cousins DJ, Lavender P, Calder VL, Lord GM, Jenner RG. T-bet Activates Th1 Genes through Mediator and the Super Elongation Complex. Cell Rep. 2016 Jun 21;15(12):2756-70. doi: 10.1016/j.celrep.2016.05.054. Epub, 2016 Jun 9. PMID:27292648 doi:http://dx.doi.org/10.1016/j.celrep.2016.05.054
- ↑ Koch MA, Tucker-Heard G, Perdue NR, Killebrew JR, Urdahl KB, Campbell DJ. The transcription factor T-bet controls regulatory T cell homeostasis and function during type 1 inflammation. Nat Immunol. 2009 Jun;10(6):595-602. doi: 10.1038/ni.1731. Epub 2009 May 3. PMID:19412181 doi:http://dx.doi.org/10.1038/ni.1731
- ↑ Szabo, S. J., Kim, S. T., Costa, G. L., Zhang, X., Fathman, C. G., & Glimcher, L. H. (2000). A novel transcription factor, T-bet, directs Th1 lineage commitment. Cell, 100(6), 655–669. https://doi.org/10.1016/s0092-8674(00)80702-3
- ↑ Lazarevic V, Chen X, Shim JH, Hwang ES, Jang E, Bolm AN, Oukka M, Kuchroo VK, Glimcher LH. T-bet represses T(H)17 differentiation by preventing Runx1-mediated activation of the gene encoding RORgammat. Nat Immunol. 2011 Jan;12(1):96-104. doi: 10.1038/ni.1969. Epub 2010 Dec 12. PMID:21151104 doi:http://dx.doi.org/10.1038/ni.1969
- ↑ 11.0 11.1 Douglas.S.Robinson and Clare M Lloyd. Asthma: T-bet - A master controller ? Volume 12, Issue 9, PR322-R324, April 30, (2002) https://doi.org/10.1016/S0960-9822(02)00830-8
- ↑ Kardan M, Rafiei A, Ghaffari J, Valadan R, Morsaljahan Z, Haj-Ghorbani ST. Effect of ginger extract on expression of GATA3, T-bet and ROR-gammat in peripheral blood mononuclear cells of patients with Allergic Asthma. Allergol Immunopathol (Madr). 2019 Jul - Aug;47(4):378-385. doi:, 10.1016/j.aller.2018.12.003. Epub 2019 Feb 10. PMID:30745246 doi:http://dx.doi.org/10.1016/j.aller.2018.12.003
- ↑ Oh S, Hwang ES. The role of protein modifications of T-bet in cytokine production and differentiation of T helper cells. J Immunol Res. 2014;2014:589672. doi: 10.1155/2014/589672. Epub 2014 May 13. PMID:24901011 doi:http://dx.doi.org/10.1155/2014/589672
- ↑ Tantisira KG, Hwang ES, Raby BA, Silverman ES, Lake SL, Richter BG, Peng SL, Drazen JM, Glimcher LH, Weiss ST. TBX21: a functional variant predicts improvement in asthma with the use of inhaled corticosteroids. Proc Natl Acad Sci U S A. 2004 Dec 28;101(52):18099-104. doi:, 10.1073/pnas.0408532102. Epub 2004 Dec 16. PMID:15604153 doi:http://dx.doi.org/10.1073/pnas.0408532102
- ↑ Pan L, Chen Z, Wang L, Chen C, Li D, Wan H, Li B, Shi G. Deubiquitination and stabilization of T-bet by USP10. Biochem Biophys Res Commun. 2014 Jul 4;449(3):289-94. doi:, 10.1016/j.bbrc.2014.05.037. Epub 2014 May 17. PMID:24845384 doi:http://dx.doi.org/10.1016/j.bbrc.2014.05.037
- ↑ Spector SL, Wangaard CH, Farr RS. Aspirin and concomitant idiosyncrasies in adult asthmatic patients. J Allergy Clin Immunol. 1979 Dec;64(6 Pt 1):500-6. doi:, 10.1016/0091-6749(79)90059-9. PMID:512268 doi:http://dx.doi.org/10.1016/0091-6749(79)90059-9