Introduction
(SMP) is a 3-subunit complex essential for cell proliferation and the survival of many cancers[1] and RASopathies.[2] The SMP holophosphatase complex functions as a key regulator of the receptor tyrosine kinase (RTK) signaling pathway by removing an inhibitory phosphate on the RAF family of proteins to allow for MAPK signaling.[2] This interaction of the RTK-Ras pathway and the SMP complex drives cell proliferation.[1] The SMP complex is made of three subunits, SHOC2, PP1C, and MRAS. Each of these subunits has a different shape that corresponds to its different function. uses a crescent shape to enhance substrate interactions and complex stability.[3] is a phosphatase, and it contains the catalytic site of the complex which dephosphorylates the N-terminal phosphoserine (NTpS) of RAF green link here.[3] binds to GTP which triggers assembly of the SMP complex. The C-terminus of the MRAS subunit localizes the complex to the cell membrane.[3] Mutations in one or multiple of these subunits leads to over-activation of the signaling pathway, which may result in cancer and developmental disorders called RASopathies.[2]
There are many regulatory mechanisms that serve as a lock on this RAS-MAPK pathway, decreasing the likelihood of unintentional pathway activation. One is a protein dimer called 14-3-3 that keeps inactive RAF localized to the cytoplasm. An N-terminal phosphorylated serine keeps RAF bound to this protein dimer, and when the SMP complex is assembled, the catalytic subunit, PP1C, removes the phosphate group from the serine, releasing RAF from the 14-3-3 dimer, and activating the RAS-MAPK cell proliferation pathway.
In all images and animations, SHOC2 will be shown as cyan blue, MRAS as lime, and PP1C as violet. Other important components involved in the function of the SMP complex include the 14-3-3 dimer and Raf, which will be shown in salmon and slate-blue, respectively.
Significance
Cell Proliferation
The Ras-Raf signaling cascade as a whole is fundamental for cell growth and survival. When a membrane bound GTPase is activated by extracellular growth proteins, it binds to a GTP molecule which then activates Raf and the signaling cascade. However, Raf is typically kept in an auto-inhibited form. When MRAS is GTP-bound rather than GDP-bound, it triggers the formation of the SMP complex. The active site of PP1C, when in complex, is responsible for removing the residue that causes steric clash, and therefore, auto-inhibition. Extracellular growth factors trigger both formation of the SMP complex and Ras-Raf interaction through the binding of GTP to a Ras-protein, however, the SMP complex must remove the auto-inhibition before Ras and Raf can interact. Since SHOC2-PP1C-MRAS plays such a crucial role in the activation of the signaling cascade, many scientists say that cell proliferation is regulated by the SMP complex rather than the Ras-Raf interaction.
Cancer and Rasopathies
Mutations in any of the 3 subunits of SHOC2-PP1C-MRAS can lead to cancer or a developmental disability called Rasopathy. Mutations occur at the protein-protein interaction surfaces, leading to more stability of the complex as well as increased interaction energy of SHOC2 with PP1C and/or MRAS.[2] For SHOC2 and PP1C, the mutations lead to amino acid changes on the interaction surfaces, causing a higher affinity for binding.[4] Mutations to MRAS lead to consistent GTP-loading, causing an increase in the formation of the SMP complex. As a result, there is consistent activation of the cell-proliferation pathway even without the presence of the external growth factors. Because the system is no longer regulated, cells proliferate regardless of external signals, leading to cancer and/or RASopathies. Furthermore, some mutations in PP1C lead to increased active site enzymatic activity, also leading to increased cell growth.
Structure of Subunits
SHOC2
SHOC2 is essential for complex formation, however SHOC2 only undergoes a 6° when MRAS and PP1C bind.[1] SHOC2 is just the place where MRAS and PP1C come together. SHOC2 and PP1C first engage in binding with each other, and MRAS-GTP binds, stabilizing SHOC2 and PP1C binding, and fully forming the SHOC2-MRAS-PP1C holophophatase complex. [2]
PP1C
The protein phosphatase 1 catalytic (PP1C) subunit contains the catalytic site of the complex which dephosphorylates the N-terminal phosphoserine (NTpS) of RAF.[3] When PP1C is bound to the surface formed between SHOC2 and MRAS, the active site is exposed and fully accessible for the substrate to bind in the holoenzyme complex.[1] PP1C cannot act independently from the SMP complex because it lacks intrinsic substrate selectivity.[3] Therefore, PP1C requires the presence of SHOC2 and MRAS to function properly, whereas SHOC2 and MRAS may interact in a binary complex without the presence of PP1C.[1] SMP complex formation is initially mediated by SHOC2 and PP1C, then stabilized by the GTP interactions in MRAS and SHOC2.[2]
PP1C activity is regulated by short linear interaction motifs or PP1C-binding regulatory proteins.[1] The regulatory proteins bind to small motifs in PP1C, like RVXF.[3] The RVXF motif is located in PP1C through the N-terminal disordered region.[2]
There is a direct interaction between the RVXF motif of SHOC2 and the RVXF-binding pocket of PP1C.[1]
SHOC2 has a RVxF binding motif that interacts with the PP1C RVxF binding site. The N-terminal loop of SHOC2 interacts with the RVxF binding site of PP1C, highlighting the structure and function connection of the complex. RVxF allows PP1C substrates to bind, whereas RAF has the RVxF motif, so it can bind to the hydrophobic region of SHOC2, allowing for greater specificity. Additionally, PP1C and SHOC2 do not change conformationally upon the binding of GTP, but rather they are inactive when RAS is bound to GDP due to steric strain. with or without binding to the SMP complex as PP1C retains its enzymatic function independently.[3].
MRAS
Ras proteins are GTP dependent switches that are associated with the plasma membrane.[3] Ras proteins often regulate cycles during signal transduction. MRAS, one of the subunits in the SMP complex, is a RAS protein specific to SHOC2 and the SMP complex. Other RAS proteins may bind to SHOC2, but MRAS induces the complex formation with a significantly lower Kd (dissociation constant), thus has the strongest connection.[3] The interface between SHOC2 and MRAS consists of two switches, Switch I and Switch II.[3] The switch regions were the only regions in MRAS to conformationally change, depending on the state of GTP.[3]
The formation of the SMP complex is stabilized and driven by the MRAS GTP-bound active state.[1][2] The tertiary structure formation is GTP dependent on multiple RAS forms.[1] When GTP is bound to MRAS, the SMP complex forms and MRAS is in the active form. When GDP is bound to MRAS, the SMP complex does not form and MRAS is in the inactive form.[3] In order for MRAS to bind to SHOC2, MRAS must be in the active GTP bound state. When the inactive GDP is bound to MRAS, steric clashes between Switch 1 on MRAS and PP1C prevent SHOC2 binding and the SMP complex formation.[3]
Additionally, the surface of MRAS that is buried in the complex overlaps the surfaces used to engage RAF, requiring two separate MRAS proteins to activate a single RAF molecule, one in the SMP complex and one to dephosphorylated Raf to activate the MAPK signaling cascade. The SMP complex is localized to the cell membrane or other RAS isoforms by the palmitoylated, C-terminus end of MRAS.[1] In its , Ras has an extended, palmitoylated C-terminal helix which allows it to bind to the cell membrane.[3]
Autoinhibited Confirmation
The first step of the signaling cascade is the dephosphorylation of Raf at Ser259. In the , Raf interacts with a 14-3-3 dimer due to the phosphate group present on Ser259. This interaction with 14-3-3 restrics Raf to the cytoplasm and inhibits Raf from binding with Ras due to steric clash. When GTP binds to MRAS, this triggers the SMP complex to form. Once the complex is formed, PP1C is brought into close proximity of Ras, leading to the dephosphorylation of Ser259. Once dephosphorylated, Raf is in the , allowing for the interaction of Ras and Raf, and the initiation of the signaling cascade.[5]
Signaling Cascade and Conformational Changes
Switch I and Switch II
SHOC2-PP1C-MRAS is a regulator of a cell proliferation pathway. Mutations in cell proliferation pathways are responsible for 25% of all cancers 1. If this cell proliferation pathway goes unmediated, rapid cell growth and division will occur; the leading cause of cancers is mutations in this pathway. [4] Mechanistic Overview and Signaling Cascade shows the pathway SHOC2-PP1C-MRAS is part of. It is a receptor tyrosine kinase pathway.[2] When MRAS is bound to GDP, the complex is not assembled. SHOC2, PP1C, and MRAS all exist as separate monomers. The Raf domain contains a kinase domain (KD), Ras binding domain (RBD), a C-terminal phosphoserine (CTpS), a N-terminal phosphorylated serine (NTpS), and a 14-3-3 protein dimer, restricting RAF to the cytoplasm. In the activated pathway, MRAS is bound to GTP, and the SMP complex is assembled. PP1C is now in contact with the NTpS, allowing it to become dephosphorylated. [4] This dephosphorylation causes the dimerization of two Raf proteins via their kinase domains as well as a conformational change. This conformation change causes the phosphorylation of other residues. Eventually, this leads to the unbinding of GDP from MRAS and the binding of GTP to MRAS, causing a shift from the to The Switch I region is made up of residues 42-48 of the MRAS domain.[2] These residues are crucial for the binding of MRAS, SHOC2, and PP1C. When GDP is bound to the MRAS domain, it is in the . Since the gamma P is not bound to GDP, there are no hydrogen bond interactions with the oxygens of the phosphate group- hence the open conformation. When GTP is bound to MRAS, it is in the . The closed conformation allows for the binding of SHOC2 and PP1C. The open conformation of MRAS sterically clashes with the binding site of SHOC2, which is why the complex is not assembled when GDP is bound. [2].
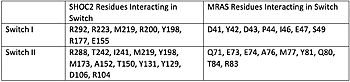
Figure 1. Residues Interacting at SWI and SWII at subunits SHOC2 and PP1C.
[3].
Switch I (SWI) and Switch II (SWII) are located between the SHOC2 and MRas subunits. When GTP is hydrolyzed to GDP, Switch I and Switch II relax, in the relaxed state SHOC2 cannot bind to MRas. Two Residues from MRas interact with the gamma phosphate on GTP, changing the complex to the closed confirmation. When GTP is bound to , it triggers the assembly of the SHOC2 Complex. When SWI is in its open confirmation, PP1C cannot bind with MRas due to the steric clashes, but when GTP binds and SWI is in its closed confirmation, PP1C can bind without hinderance. In a mutated complex, other RAS proteins can replace MRas making cell proliferation more likely. SHOC2-PP1C-MRas may be used as a therapeutic target for cancer treatments through changing the confirmation of the .
Ras/Raf
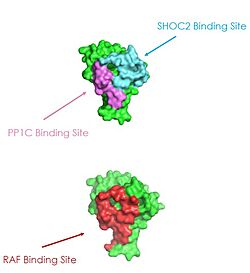
Figure 2: MRAS binding sites with SHOC2, PP1C, and RAF.
[3].
Ras proteins are GTP-dependent intracellular switches that are anchored to the plasma membrane, which activate RAF kinases through direct binding and membrane recruitment, resulting in RAF dimerization and pathway activation. [3]. Ras has a hydrophobic fatty acid tail, keeping it anchored to the membrane. There are no known membrane interacting regions on SHOC2 and PP1C, meaning MRAS likely recruits them to the membrane. As seen in these figures, there is a significant amount of steric overlap with MRAS binding site with PP1C and SHOC2 and Raf. Hence, multiple Ras proteins are required for further activation of the receptor tyrosine kinase pathway. One Ras molecule is needed to recruit SHOC2 and PP1C to the membrane, and one Ras molecule is needed activate Raf. The ability of Ras-GTP to cluster at the membrane is a crucial capability for this protein complex. This anchoring is possible due to the presence of a hydrophobic fatty acid tail on Ras. One RAS molecule is needed to recruit SHOC2 and PP1C to the membrane, and one RAS molecule is needed activate Raf.
Structure of Active Site
3-Metal Ion Catalysis
The of the SHOC2-PP1C-MRAS complex resides in the PP1C subunit.[6] The role of PP1C is to dephosphorylate SER259 of Raf so that the signaling cascade can start. The active site is unchanged upon the binding of the complex, however, SHOC2 and MRAS aid in the specificity of the enzymatic activity as PP1C is able to dephosphorylate many different targets on its own, with almost 100 PP1C targets found.[5] The full mechanism for the catalytic activity is unknown, however, there are 3 metal ions present (2-Mg2+ and 1-Cl-) to stabilize the waters present in the active site. Additionally, the substrate binds through hydrogen bonds with the main chain and side chain atoms of the catalytic residues. Mutations in the active site lead to increased activity, causing the Ras/Raf signaling cascade to be triggered more frequently.[6]
Hydrophobic Binding Site
PP1C has a
adjacent to its active site.[6] The majority of PP1C targets are able to bind through a specific motif that is recognized by the hydrophobic groove. In the Ras/Raf signaling cascade, the region of Raf that is C-terminal to the phosphate group binds to the hydrophobic groove, and the remaining residues bind to the hydrophobic region of SHOC2. This binding to SHOC2 is what allows the SMP complex to be more specific than PP1C on its own.[6] PP1C also has a singular cysteine (C291) present in the hydrophobic binding site in order to provide further stability to the substrate-protein interaction.
Future Directions
The knockdown of SHOC2 is being studied as a target for cancer and RASopathy treatment.[2] Although MRAS is the protein that triggers the formation of the complex, SHOC2 is the anchoring location for both MRAS and PP1C. Without SHOC2, the complex would not form and SER259 would not be dephosphorylated. MRAS could be triggered and moved towards the cell membrane, but no complex will form and Raf will remain in the auto-inhibited form. Additionally, there are other RAS proteins that can form an SMP-like complex. If MRAS were to be depleted, other RAS proteins could step in place of MRAS. PP1C is able to dephosphorylated other proteins on it's own, therefore it is not a good target as depletion of PP1C could lead to other issues. Depletion of SHOC2 is the most promising treatment that has been researched. There is also possibility that changing the confirmation of RAS Switch II could lead to decreased cell proliferation.


