Introduction
![Figure 1: An overview of the Thyroid System. A depiction of signaling cascade from the hypothalamus ending in the release of TSH causing T3 and T4 production and its effects. The mechanism of regulation is also shown by negative feedback from the T3 and T4 hormones. Source: [1]](/wiki/images/thumb/1/1d/TSH_system1.png/300px-TSH_system1.png)
Figure 1: An overview of the Thyroid System. A depiction of signaling cascade from the hypothalamus ending in the release of TSH causing T3 and T4 production and its effects. The mechanism of regulation is also shown by
negative feedback from the T3 and T4 hormones. Source:
[1]Thyroid Stimulating Hormone Receptor (TSHR) is a G-Protein Coupled Receptor (GPCR) found in human thyroid follicles[1]. TSHR is activated by the Thyroid Stimulating Hormone (TSH) also known as thyrotropin. Activation of TSHR initiates a signaling pathway for the production of thyroid hormones such as T3 and T4 that are used to regulate the metabolism(Fig 1).
Structure
TSHR forms an active signaling complex with TSH and Gs proteins. This is called the . TSH contains an α and a β subunit. The α subunit is a shared subunit amongst glycoproteins. The β subunit is unique to TSH. TSH binds to the extracellular domain of TSHR [2].
: Leucine Rich Region Domain (coral), the hinge region (blue-purple), and the transmembrane region(rainbow). The leucine-rich region domain is the extracellular TSH ligand domain. The hinge connects the Leucine Rich Repeat Domain and the Transmembrane Region. It provides flexibility for the switch between the active and inactive state of TSHR. The transmembrane region is located within the plasma membrane. Its function transmits the extracellular signal across the membrane to the intracellular G-proteins bound to the N-terminus of the transmembrane region[2]. Activated G-proteins then signal a robust intracellular signaling cascade.
Leucine Rich Domain
The is the extracellular ligand binding region of TSHR. Connects to the Hinge Region at its C-terminus. It is made up of polar residues in a concave parallel β-sheet composed of leucine-rich repeats where TSH binds and is called the [2]. The LRRD concave surface is where the TSH antibody antagonist, K1, binds as well as the agonist M22. Two specific LYS residues interact with the substrate to result in a structural change of the molecule. N glycans attached to asparagine residues play a large role in the binding of TSH. Negative charge on these N glycans contributes to the polarity of the binding pocket which mediates the binding efficiency of TSH. TSHR can be glycosylated at residues 77, 99, 113, 177, 198, and 302[3]. Four of the five N glycan sites must be glycosylated for TSHR to be in the active form[3].
Hinge Region
The (purple-blue) connects the Transmembrane Region to the Leucine Rich Domain. Also referred to as the signaling specificity domain the hinge region plays a dual role in both TSH binding and signal transduction. [4]. The hinge region is made up of two α-helices connected via disulfide bonds. The disulfide bonds are important for transmitting the signal to the Transmembrane region. The two helices help orient TSH properly for LRRD binding. It is proposed that the Asp386, Tyr385, and Tyr387 create a negative-charged region on the Helix. This negatively charged region interacts with the positively charged region of TSH created by residue Arg54. These interactions are essential for TSH binding, however, they are not required for the activation of TSHR. This is known because the M22 antibody is able to activate TSHR even though it does not have an Arg54 residue Conformational changes in this hinge region, specifically the orientation of , which is located in the hinge region, are responsible for the bringing TSHR into the active state [1].
Transmembrane Region
() is embedded within the cell membrane, like other G-protein receptors, it is composed of a 7-pass helix [1]. The transmembrane region is surrounded by a "belt" of . When cholesterol binding sites are mutated, TSHR activity decreases. These cholesterols are likely important for TSHR function [2]. Additionally, at the N-terminus, the transmembrane region binds to the , which are located intracellularly [5]. The G-proteins are made up of three subunits: α, β, and γ. Nb35 a fourth subunit that stabilizes the structure by binding between the Gα and Gβ interface[6]. When TSHR is activated, it causes the Gα subunit to dissociate from the Gβγ subunits. The Gα subunit is responsible for activating adenylyl cyclase, phospholipase C, and ion channels. This sets off the robust intracellular signaling cascade[5].
Antibodies
Antibodies are important for the thyroid response to regulate the metabolism. These proteins are made from an immune system response to get rid of unwanted antigens in the body. Thyroid antibodies are made when the body attacks the thyroid tissues. These antibodies are made to mimic TSH (Thyroid Stimulating Hormone). TSH is released from the pituitary gland to bind to TSHR and stimulate the thyroid to make T3 and T4 hormones to regulate the metabolism. These thyroid antibodies bind to the concave surface of the LRRD.
M22 and K1
M22 is an activating antibody for TSHR. This antibody mimics TSH to activate the thyroid gland to produce T3 and T4 hormones. M22 makes a stronger interaction with TSHR than TSH does due to a larger number of hydrogen bonds (14 H bonds for M22 and 3 for TSH) and salt bridge interactions with the concave surface of the LRRD[7]. This interaction is key for understanding why M22 activates TSHR and does not release TSHR to go into the inactive state even when T3 and T4 levels are high[7].
K1 is an inhibitory antibody. This antibody mimics TSH and binds to TSHR with a high affinity which prevents the receptor binding to TSH. K1 puts TSHR in the inactive conformation[8]. It is effective at lowering T3 and T4 levels while increasing TSH concentration.
Active vs. Inactive State
In its resting state without TSH bound, TSHR is in the , also known as the "down" state because the LRRD is pointing down. When TSH binds to TSHR, steric clashes between TSH and the cell-membrane cause TSHR to take on the (Fig 2). During this transition, the LRRD rotate 55° along an axis perpendicular to the cell membrane. This rotation is initiated by conformational changes within the , specifically at the , located in the Hinge Region. Y279 moves 6 Å relative to I486, which is a residue located in the Transmembrane Region nearby Y279[1] (Fig 3).
The active form is favored when . The structure can be seen as straight. The same straight conformation is observed when TSHR is bound with M22. The inactive form is found when of TSHR is found when bound with K1. The overall structure of the molecule is bent when K1 is bound.
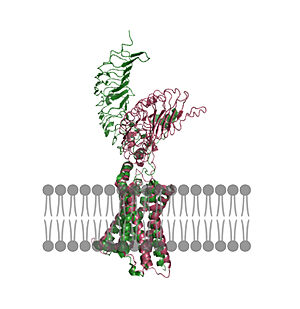 Figure 2: An overview of the Inactive (pink) vs Active (green) state of TSHR embedded in the plasma membrane. In the inactive state, the LRRD is pointed down. When TSH binds to the LRRD, conformation changes in the Hinge Region and steric clashes between TSH and the Cell membrane cause TSHR to rotate into the active state. As shown, the LRRD rotates 55° into an upright position [1]. PDB: 7t9m and 7t9i. | 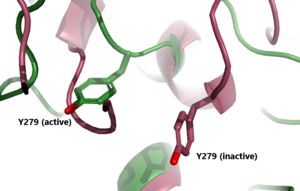 Figure 3: A zoomed in view of the Y279 residue in the Hinge Region of TSHR, showing the 6Å rearrangement of Y279 during the activation of TSHR. Active TSHR is shown in green (PDB: 7t9i) and inactive TSHR is shown in pink (PDB: 7t9m). |
Specific Residues and Interactions
On the concave surface of the LRRD two are the main contributors to the binding of the antibodies. The concave structure of the binding pocket allows a with antibodies. Specifically, interacts with Glu 118 and interacts with Asp 111 on K1 and M22 antibodies to make a salt bridge interaction. The interaction is not close enough to make a hydrogen bond. Instead, the interaction between the Lys residues with the Asp or Glu residues is a salt bridge interaction because the distance between the residues is greater than 2Å. When in the inactive form, LYS 209 interaction is absent but LYS 58 interaction with Glu 118 is present. These salt bridge interactions are highly specific as mutation of Lys209 to Arg induced a different interaction. This new interaction widens the selectivity of TSHR, allowing it to bind to other hormones[9].
Biological Relevance
![Figure 4: T3 and T4 role in TSH concentration: Highlighting the problem when under or overactive on the metabolism. When an antibody is bound to TSHR and cannot respond to the negative feedback look the metabolism experiences a shift outside of equilibrium resulting in a wide array of side effects. [2]](/wiki/images/thumb/5/5f/T3t4levels.jpeg/400px-T3t4levels.jpeg)
Figure 4: T3 and T4 role in TSH concentration: Highlighting the problem when under or overactive on the metabolism. When an antibody is bound to TSHR and cannot respond to the negative feedback look the metabolism experiences a shift outside of equilibrium resulting in a wide array of side effects.
[2]The thyroid plays an essential role in the body's metabolism functions, including heart rate, digestion, and temperature regulation. When TSH is bound to TSHR, a signal is sent to the thyroid to produce the thyroid hormones, T3 and T4.
T4 is an inactive form until converted into T3. T3 and T4 are transmitted through the body to increase or decrease your metabolism. T3 and T4 then use a feedback mechanism to regulate the release of TSH by the pituitary gland(Fig.4). High T3 and T4 levels inhibit TSH production. Whereas low levels permit TSH binding to TSHR.
Hyperthyroidism
Hyperthyroidism is when the thyroid is overactive which leads to upregulation of the metabolism. This overactive state can be initiated by antibodies like M22. M22 antibody maintains TSHR in the active conformation. In this conformation, the thyroid continues to make T3 and T4, overstimulating the metabolism. Symptoms of hyperthyroidism include fast or irregular heartbeats, tiredness, increased hunger, sleep problems, enlarged thyroid gland, and sensitivity to heat. Grave's Disease is the most common cause of hyperthyroidism. This is an autoimmune disorder that causes your body to attack the thyroid gland[10]. When someone has Hypothyroidism the body produces M22 that leads to overactive metabolism functions.
Hypothyroidism
Hypothyroidism is when the thyroid is underactive leading to a decrease in metabolism function. K1 antibody binds to TSHR to promote the inactive confirmation that is conserved despite the presence of TSH. This does not allow for the signaling of the T3 and T4 hormones to upregulate the metabolism. The symptoms of this include slow or irregular heartbeats, tiredness, muscle aches, memory problems, jaundice, and sensitivity to cold. Hasimoto's disease is an example of hypothyroidism. This is an autoimmune disorder that causes your body to attack the healthy cells of the thyroid. Specifically causing the death of the cells that produce the thyroid hormones. When the thyroid fails to produce its hormones it activates TSH production through a negative feedback mechanism[10]
![Figure 1: An overview of the Thyroid System. A depiction of signaling cascade from the hypothalamus ending in the release of TSH causing T3 and T4 production and its effects. The mechanism of regulation is also shown by negative feedback from the T3 and T4 hormones. Source: [1]](/wiki/images/thumb/1/1d/TSH_system1.png/300px-TSH_system1.png)


![Figure 4: T3 and T4 role in TSH concentration: Highlighting the problem when under or overactive on the metabolism. When an antibody is bound to TSHR and cannot respond to the negative feedback look the metabolism experiences a shift outside of equilibrium resulting in a wide array of side effects. [2]](/wiki/images/thumb/5/5f/T3t4levels.jpeg/400px-T3t4levels.jpeg)
