This is a default text for your page '. Click above on edit this page' to modify. Be careful with the < and > signs.
You may include any references to papers as in: the use of JSmol in Proteopedia [1] or to the article describing Jmol [2] to the rescue.
Introduction
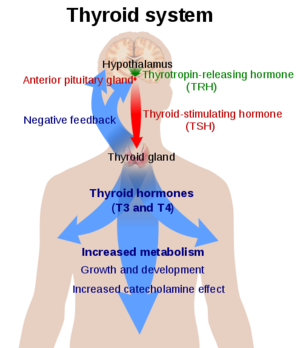
Thyroid Stimulating Hormone Receptor (TSHR) is a G-Protein Coupled Receptor (GPCR) found in human thyroid follicles. TSHR is activated by the Thyroid Stimulating Hormone (TSH) also known as thyrotropin. Activation of TSHR is initiates a signaling pathway for the production of thyroid hormones such as T3 and T4 (Fig 1).
Structure
TSHR forms an active signalling complex with TSH and Gs proteins. This is called the . TSH contains an α and a β subunit. The α subunit is a shared subunit amongst glycoproteins. The β subunit is unique to TSH. TSH binds to the extracellular domain of TSHR [3].
: Leucine Rich Region Domain (coral), the hinge region (blue-purple), and the transmembrane region(rainbow). The leucine rich region domain is the extracellular TSH ligand domain. The hinge connects the Leucine Rich Repeat Domain and the Transmembrane Region. It provides flexibility for the switch between the active and inactive state of TSHR. The transmembrane region is located within the plasma membrane. Its function transmit the extracellular signal across the membrane to the intracellular G-proteins bound to the N-terminus of the transmembrane region[3]. Activated G-proteins then signal a robust intracellular signaling cascade.
Transmembrane Region
() is embedded within the cell membrane, like other G-protein receptors, it is composed of a 7-pass helix [4]. The transmembrane region is surrounded by a "belt" of [3]. When cholesterol binding sites are mutated, TSHR activity decreases. These cholesterols are likely important for TSHR function [3]. Additionally, at the N-terminus, the transmembrane region binds to the , which are located intracellularly [5]. The G-proteins are made up of three subunits: α,β, and γ. When TSHR is activated, it causes the Gα subunit to dissociate from the Gβγ subunits. The Gα subunit is responsible for activating adenylyl cyclase, phospholipase C and ion channels. This sets off the robust intracellular signaling cascade[5].
Leucine Rich Domain
The is the extracellular ligand binding region of TSHR. It is made up of about 280 different residues. Connected to its C-terminus is the Hinge Region. It is made up of an extensive parallel β-sheet. This β-sheet is where TSH binds and is called the binding pocket [3].
Hinge Region
The (purple-blue) connects the Transmembrane Region to the Leucine Rich Domain. Also referred to as the signaling specificity domain the hinge region plays a dual role in both TSH binding and signal transduction. [6]. The hinge region is made up of two α-helices connected via di-sulfide bonds. The disulfide bonds are important for transmitting the signal to the Transmembrane region. The two helices help orient TSH properly for LRRD binding. It is proposed that the Asp386, Tyr385, and Tyr387 create a negative-charged region on the Helix. This negatively charged region interacts with the positively charged region of TSH created by residue Arg54. These interactions are essential for TSH binding, however, they are not required for the activation of TSHR. This is known because the M22 antibody is able to activate TSHR even though it does not have an Arg54 residue Conformational changes in this hinge region, specifically the orientation of , which is located in the hinge region, are responsible for the bringing TSHR into the active state [4]
Active vs Inactive State
In its resting state without TSH bound, TSHR is in the , also known as the "down" state because the LRRD is pointing down. When TSH binds to TSHR, steric clashes between TSH and the cell-membrane cause TSHR to take on the (fig 2). During this transition, the LRRD rotate 55° along an axis perpendicular to the cell membrane. This rotation is initiated by conformational changes within the , specifically at the , located in the Hinge Region. Y279 moves 6 Å relative to I486, which is a residue located in the Transmembrane Region nearby Y279[4] (Fig 3).
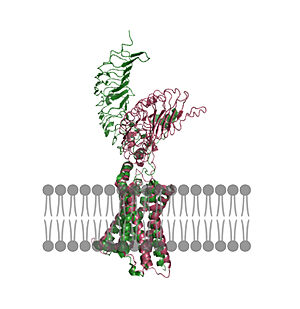 Figure 2: An overview of the Inactive (pink) vs Active (green) state of TSHR embedded in the plasma membrane. In the inactive state, the LRRD is pointed down. When TSH binds to the LRRD, confirmation changes in the Hinge Region and steric clashes between TSH and the Cell membrane cause TSHR to rotate into the active state. As shown, the LRRD rotates 55° into an upright position [4]. PDB: 7t9m and 7t9i. | 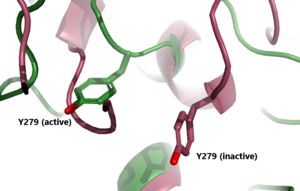 Figure 3: A zoomed in view of the Y279 residue in the Hinge Region of TSHR, showing the 6Å rearrangement of Y279 during the activation of TSHR. Active TSHR is shown in green (PDB: 7t9i) and inactive TSHR is shown in pink (PDB: 7t9m). |
Specific Residues
Biological Relevance
This is a sample scene created with SAT to by Group, and another to make of the protein. You can make your own scenes on SAT starting from scratch or loading and editing one of these sample scenes.