This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
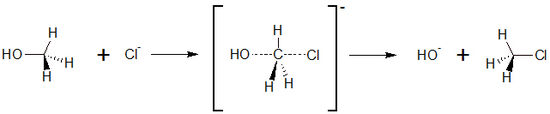
Sandbox testing Sn2 for Veronika
From Proteopedia
Your Heading Here (maybe something like 'Structure')
| |||||||||||
The animation was originally done by Prof. Dr. Verena Pietzner; for details, see her web site ChiLe[2]. The implementation into Proteopedia was done by Prof. Jaime Prilusky, Prof. Joel L. Sussman and Veronika Pelekhov.
See also
SN1 reaction: Substitution of Cl− and tert-Butanol
References
- ↑ Wang Y, Song H, Szabo I, Czako G, Guo H, Yang M. Mode-Specific SN2 Reaction Dynamics. J Phys Chem Lett. 2016 Sep 1;7(17):3322-7. doi: 10.1021/acs.jpclett.6b01457. Epub, 2016 Aug 12. PMID:27505286 doi:http://dx.doi.org/10.1021/acs.jpclett.6b01457
- ↑ ChiLe Web Site