This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Induced fit
From Proteopedia
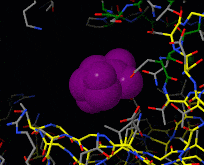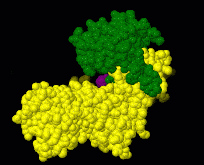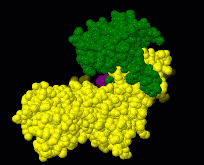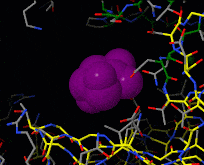
Induced fit describes a conformational change in a protein when it binds a ligand, in contrast to a lock-and-key model of ligand binding. A classic example of induced fit is binding of glucose to hexokinase, depicted in a morph between 3o8m and 3o80 in the picture at right.
Contents |
History of the concept
Induced fit was suggested by Koshland in 1958 [1], providing an alternative to the lock-and-key binding model that Emil Fischer proposed in 1899 [2].
Interactive examples
| |||||||||||
See Also
- A morph of induced fit when Tamiflu binds to Neuraminidase.