Introduction
Stearoyl-CoA Desaturase is an enzyme essential for the biosynthesis of monosaturated fatty acids from saturated fatty acids [1]. SCD catalyzes the rate-limiting step in the conversion of Stearoyl-CoA to oleic acid, an essential substrate in the biosynthesis of phospholipids, triacyclglycerols, and cholesterol [2]. SCD is highly conserved in eukaryotes and has different gene isoforms. Mice have four isoforms: SCD1, SCD2, SCD3, and SCD4. Humans have two different isoforms: SCD1 and SCD5 [2]. The SCD isoform discussed in this page is Stearoyl-CoA Desaturase 1 (SCD1) found in mice. SCD is believed to have once been an anaerobic pathway found in cartilaginous fish about 450 million years ago [3]. The enzyme’s mechanism is now aerobic and favorable. The structure of SCD1 was found using X-ray crystallography [2].
Structure
Overall Structure
SCD1 is an integral membrane protein embedded within the endoplasmic reticulum and consists of 4 transmembrane alpha helices, arranged in a cone-like shape. The cytosolic domain of the enzyme consists of 11 alpha helices and contains the carboxy and amino termini [2]. Its substrate, Stearoyl-CoA, binds to the cytosolic region which contains a "kink" that properly orients Stearoyl-CoA to undergo a dehydrogenation reaction between the of Stearoyl-CoA [2].
Binding of Substrate
Stearoyl-CoA is the substrate that binds to the enzyme, SCD1. The binding of the substrate is stabilized by specific residues on the exterior and interior of the protein. Stearoyl-CoA is a long-chain fatty acyl-CoA. The head group of the substrate is composed of an adenine, ribose, phosphate groups, and polar atoms such as of nitrogen, oxygen, and sulfur. The head of stearoyl-CoA is attached to the exterior of the protein by polar residues. The adenine, ribose, and phosphate are attached by the residues . The remaining exterior of the substrate is attached by the residues [2]. All the conserved residues are attached to the Stearoyl-CoA via hydrogen bonds. The fatty acid tail of Stearoyl-CoA is a 17-carbon chain which reaches into the interior of the protein. The fatty acid chain dives into the interior hydrophobic tunnel which is long, narrow, and approximately 24 Angstroms long [2]. The geometry of the tunnel and formation of bound acyl chain are the structural basis for the stereospecificity of the desaturation reaction [2].
Kink of Substrate
The chain is kinked at where the double bond is generated. The kink is induced through the interactions of four conserved residues. Three out of four of these residues are not bound to the chain, but are hydrogen bonded to each other: . T257 is hydrogen bonded to Q143, and Q143 is hydrogen bonded to W149 [2]. These residues are directly below the kink and will be hydrolyzed when the enzymatic product is ready to be released. Specifically, if the hydrogen bond between T257 and Q143 is broken, a large opening would allow for the product to be released into the bilayer [2]. The residue that is directly hydrogen bonded to the chain is . This residue is highly conserved and stabilizes the chain so it will be in the correct orientation in the active site. The enzyme will be effective on acyl chains that are between 17 to 19 carbons long. The residue that has a role in determining substrate length is . Y104 is a capping residue that has approximately 4 Angstroms between its' hydroxyl oxygen and the end of the chain [2].
Active Site
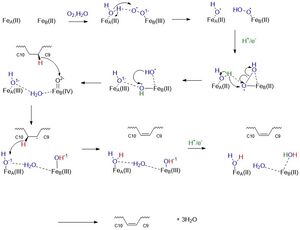
Mechanism
Disease