Introduction
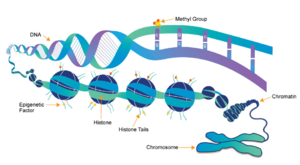
Figure 1: Epigenetic overview. Showing the methylation of histones and how this affects DNA packing.
[1]Histones are a family of proteins that condense DNA into chromatin, and is an octamer composed of two of each protein core; H2A, H2B, H3, and H4. Histones are a globular protein, that have N- or C-terminal tails. These tails can be subjected to modifications by enzymes. Methylation of histones is one of the four common histone modifications. Methylation is most common on long tails of H3 and H4 due to the tail being able to enter the active site.[2] A histone can be mono-, di-, or tri- methylated. Once the histone is methylated, the DNA goes from tightly bound heterochromatin to loosely packed euchromatin shown in Figure 1. The euchromatin allows RNA pol II to bind to the DNA and start transcription. [3] Histone methylation is a major epigenetic marker, which can be passed down from generation to generation. An epigenetic marker affects the way that genes are expressed, and can activate or repress DNA. Histone methylation is a major epigenetic marker because it has the ability to change heterochromatin to euchromatin, and vice versa. Alterations in markers have been associated with many diseases.[4] Lysine Methyltrasferase SET7/9 (KMT) is an enzyme that methylates the histone 3 lysine 4 (H3K4) and plays a important role in the transcription of DNA in Homo sapiens.[5] The methylation of H3K4 results in transcriptional activation.[5] The specific methylation of H3K4 does not result in a change in charge because it is a nonpolar group being added to the lysine. A change in charge could result in tighter bound heterochromatin.
Structure
The human lysine methyltransferase(HKMT) SET7/9 is 366 amino acids long that comes from H. sapiens. Structure has two proteins in the asymmetric unit, yet acts as a monomer. HKMT will crystalize as a dimer. The structure is composed of the ΔSET7/9 domain. There is a two-domain architecture containing a conserved anti-parallel 𝛃-barrel and an unusual knot-like structure that creates the active site. It also contains a cofactor, SAM, that plays a role in the active site.[5] HKMT only monomethylates H3K4, because the tyrosine rich active site does not allow for di- or tri- methylation.
SET Domain
The ΔSET7/9 consists of the SET domain along with the pre- and post-SET regions.[6][7] The pre- and post-SET regions are adjacent to SET domain and are cysteine rich.[6][7] The pre-SET cysteine region is located near the N-terminal where the post-SET region is located near the C-terminal of the domain.[6][7] These regions are said to play an important role in substrate recognition and enzymatic activity.[6][7] The cysteine regions are not shown in the crystal structure as only residues 117-366 were crystalized.[4]
The SET domain is mostly defined by turns and loops, which connect secondary structures, with the few .[6] form a β-hairpin that sticks out at a right angle to the surface of the enzyme.[4] This hairpin stabilizes the conformation of tyrosine residues.[4] The following three residues () accommodate a unique sharp bend in the peptide chain and the end of the protein takes on an .[4] This alpha helix packs against a beta sheet and orients the cofactor towards the active site.[4] The two most defining features of the SET domain are the C-terminal tyrosine and the . These two components have been recognized to be essential for S-adenosyl-L-methionine(SAM) binding and catalysis, which is shown as (SAH) in the structure after methylation of the histone.[6] [7] [8] The knot-like fold contains the binding sites for the cofactor SAM and the peptide substrate.[9]
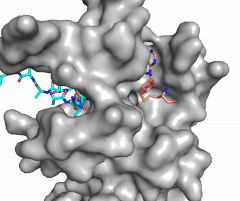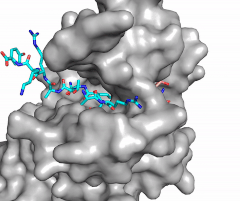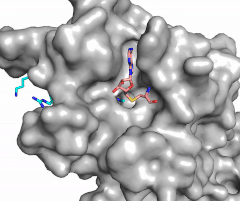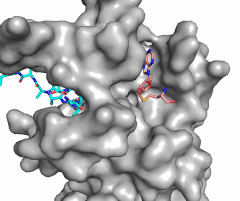
Figure 2: Image of substrate bound on one side of SET7/9 (PDB 1o9s) with the lysine target in the active site channel (cyan) and S-adenosyl homocysteine (pink) bound on the opposite face of the enzyme
Active Site and Channel
The most notable feature of the HKMT is the presence of the lysine access channel as the active site. The cofactor and substrate are located on opposite sides of the SET domain but are connected through this narrow channel (Figure 2).[4] This channel allows these two components to interact and complete the methyltransfer. The active site in general is considerably tyrosine rich. Residues Tyr245, His297, Ser268, Tyr305, Tyr335, and Tyr337 all help to shape the and the channel.[4] The cofactor involved, SAM, provides the methyl for methylation of the lysine on its sulfur atom.
The beta hairpin stabilizes the , while also shaping one side of the channel which the peptide binds to.[4] The peptide binding site is composed of residues 255-268.[4] Lysine would have trouble coming down into the active site in its charged form, but it is enabled by the faces of the flanking tyrosines.[4] The lysine is oriented so that the amine-methyl bond is pointed towards the sulfur on SAM so that it can provide the methyl. There is an important in the active site that accepts a hydrogen bond from the lysine substrate, shifting the lone pair on the nitrogen towards the sulfur of SAM. [4]
Function
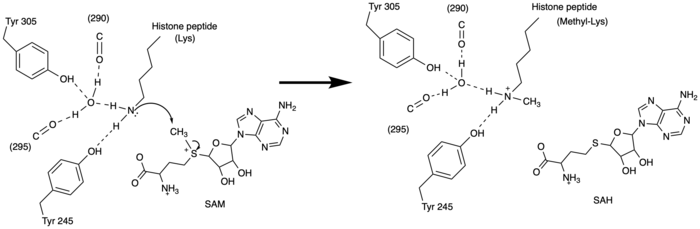
Figure 3: Histone Methylation by HKMT Mechanism
The N of the amine from the lysine serves as a nucleophile that attacks the electrophilic CH3 that is present in the AdoMet (SAM). The sulfur that the CH3 is attached to pulls the electrons towards itself to weaken the bond between the sulfur and the carbon. This weak bond allows for the N to break that bond and take the methyl group. The N on the lysine is being stabilized by Tyr residues and a water molecule (Figure 3). This allows the N to accept the methyl and take up that positive charge.
The tyrosine conserved regions of KMT have been found to be vital for only monomethylation. Multiple studies have found that a mutation to any of the conserved tyrosine residues does still monomethylate SAM, but it also creates di-methylation and tri- methylation of SAM.[10] In a recent study, Y245A and Y305F were created through site-directed mutagenesis.[10] Due to size, Ala245 was found to create a larger opening of the channel than tyrosine, which allowed for further methylation of SAM. [10] However, Y305F also showed the same characteristics of di- and tri-methylation, most likely due to a decrease of tyrosine residues shaping the narrow channel.[10] As there are four invariant tyrosine residues (Tyr305, Tyr245, Tyr335, Tyr337) in the active site, this finding indicates that the function of KMT is dependent on the presence of tyrosine residues in the active site. [10] Although only two of the tyrosines are interacting in the mechanism (Figure 3), the other two are shaping the narrow channel. Without these extra tyrosines, Tyr335 and Tyr337, the channel is not as narrow and can therefore di- and tri- methylate lysine.
Relevance
Renal Fibrosis
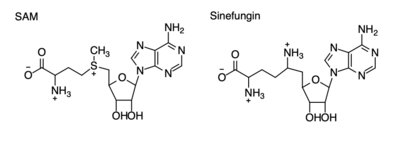
Figure 4: Structural differences between SAM and Sinefungin
The inhibition of KMT SET 7/9 has been found to enhance renal fibrosis.[11] H3K4 methylation activates the transcription of fibrotic genes, and the suppression of the H3K4 methylation was found to enhance renal fibrosis in a mouse model.[11] Sinefungin, a competitive methyltransferase inhibitor, binds to KMT to inhibit SAM.[11] SAM and Sinefungin have similar structures (Figure 4), differing only with the removal of a sulfide and the replacement of a methyl to an amine. The amine replaces the methyl donor for the reaction of KMT, inhibiting the reaction and preventing methylation. Without the methylation of H3K4, the transcription of fibrotic genes is deactivated, leading to renal fibrosis.[11]
Cancer
Many studies have shown that lysine methyltransferases lead to the inhibition of cancers, therefore they are being studied as possible cancer therapy treatments. Lysine methylation contributes to the inactivation of a tumor suppressor gene. Due to this, KMTs are being studied as possible biomarkers for the detection of cancers. Inhibitors of KMT, such as Sinefungin (Figure 4), were used in a study to observe KMT’s effect on cancerous cells. It was found that when KMT is deregulated, tumor behavior increases due to a lack of methylation, indicating the importance of the histone methylation. [12]




