This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
User:Jakob Raphael Käppler/Sandbox218
From Proteopedia
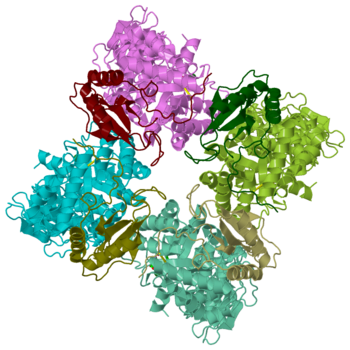
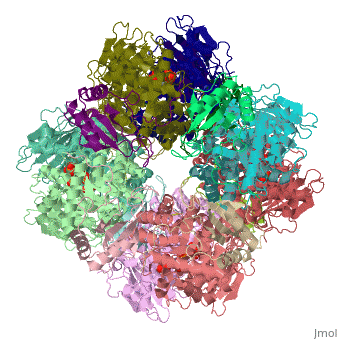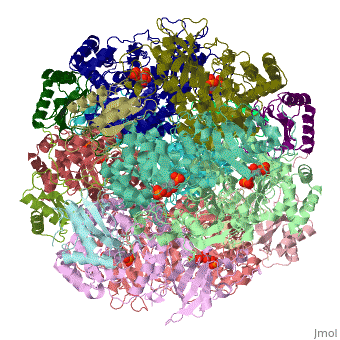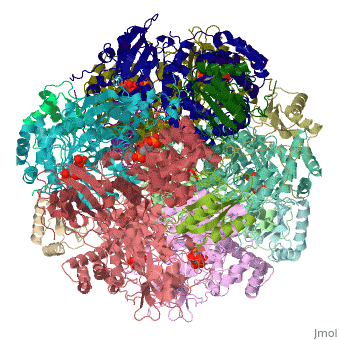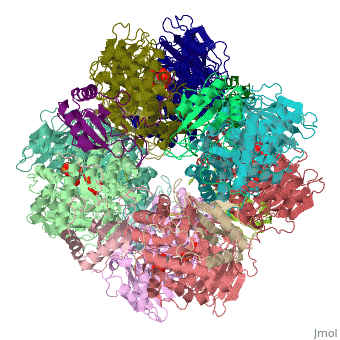
Rubisco (Ribulose-1,5-bisphosphate carboxylase oxygenase) EC 4.1.1.39, quite likely the most frequently found enzyme of the world [2] , appears in plants as well as bacteria. It simplifies the first step in the Calvin-Benson-bassham-cycle by a condensation reaction, where inorganic carbon dioxide binds in catalysis to Ribulose-1,5-bisphosphate. The enzyme, a hexadecamer with the molecular mass of 550 kDa, consists of 16 subunits (L8S8) which are divided into 8 large 50 – 55 kDa subunits and 8 small 12 – 18 kDa subunits. The genes which encode for the large subunits are located in the chloroplast whereas the genes for the small subunits are located in the nucleus. Rubisco not only catalyze the carbon fixation, it also has an oxygenase reaction. These two reactions are in competition to each other.
Contents |
Biological role
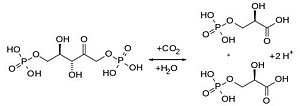
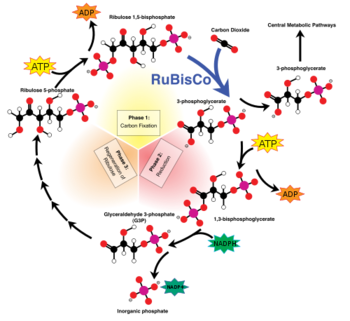
RuBisCO which is found in chloroplasts has the important biological role to catalyse the carbon fixation using CO2. The latter binds to one molecule of ribulose-1,5-bisphosphat and produces an unstable C6 (2-caboxy-3-keto-D-arabinitol-1,5-bisphosphat) intermediate product that is converted into two 3-D-phosphoglycerate. The 3-D-phosphoglycerates are used in the second step of the Calvin-cycle to build hexoses.
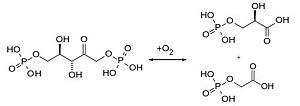
Another reaction of RuBisCO is the oxygenase reaction, where instead of carbon dioxide, O2 binds to ribulose-1,5-bisphosphate and produces an organic hydroperoxide intermediate product, that is converted to one molecule phosphoglycolate and one 3-D-phosphoglycerate[4]. In case of an oxygenase reaction, the organism uses a salvage pathway to recover the phosphoglycolate waste.
The Carboxylase/Oxygenase reactions of RuBisCO are depending on each other since CO2 is a competitive inhibitor of the oxygenase reaction and vice versa. Under normal conditions (25°C) with atmospheric CO2 and O2 concentrations the relation between oxygenase and carboxylase is 0,4 : 1 [6].
Structure
general structure
Rubisco (ribulose-1,5-bisphosphate carboxylase/oxygenase) is a large enzyme complex with a hexadecameric structure. More precisely, this enzyme has a type I structure in spinach [7]. It is composed of eight large catalytic subunits (L8, 55kDa) associated to eight smaller subunits (S8) responsible for stability [8] [6] There are four types of rubisco structures known (I, II, III and IV), but at least a third of their sequence is conserved [9]. All rubisco enzyms show similar L subunit structures. The large subunits are found in a head to tail link (α helix towards a β strand) of L2 dimers. The latter is repeated four times ((L2)4) [9]. More precisely, a single subunit L is composed of 475 amino acids [9], where 150 out of them represent the N-terminal domain and 320 amino acids compose the C-terminal domain [9]. The small subunits S (13 – 17kDa) gather 123 residues [8] and vary between the four types of rubisco enzyms known (I, II, III and IV).
active site
Rubisco is a metallo enzyme coordinated around a Mg2+ cation. In active enzymes, lysine 201 and 175, together with aspartic acid 203, glutamic acid 204 and three water molecules coordinate around the metallic cation [10].
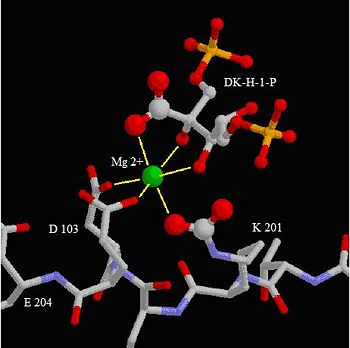
The active site in rubisco coordinates amino acids around a magnesium ion. In each four subunits the amino acids lysine 201, aspartic acid 103, glutamic acid 204 and diketo-lysine-1-phosphate interact in proximity with a magnesium ion.
Reaction
Rubisco is a bifunctional enzyme which catalyses both carboxylation and oxygenation [9]. The most important reaction of rubisco is CO2 binding. The latter is responsible for enzyme activation [9]. After binding CO2 at Lysine 201 residue in most Rubisco types and Mg++ stabilization, this enzyme forms a carbamate intermediate[9]. First RuBP, then both CO2 and water fix themselves in few steps to this molecule [9]. The reaction leads to two 3-phosphoglycerate molecules. Lysine residues in position 175 and 334 in spinach plants play a role during the catalysis [12].
Some inhibitors
The complex chemistry involved in Rubiscos’ catalysis naturally leads to diverging reactions. One of those inhibitors is xyulose-1,5-biphosphate [9]. Another one is 2-phosphoglycolate which is the result of a RuBP oxygenization. Photorespiration transforms it back to 3PGA [9].
3D Structures of RuBisCO
Updated December 2011
RuBisCO
3rg6 – RBCO – Synechococcus elongatus
3qfw - RBCO large subunit – Rhodopseudomonas palustris
1uzh, 1gk8 – CrRBCO – Chlamydomonas reinhardtii
1uw9, 1uwa – CrRBCO (mutant)
1svd – RBCD – Halothiobacillus neapolitanus
1bxn – RBCO – Cupriavidus necator
1aus - spRBCO – spinach
1rba - RrRBCO (mutant) – Rhododpirillum rubrum
5rub - RrRBCO
2wvw – RBCO – Anabena – Cryo EM
2vdh, 2vdi, 2v67, 2v68, 2v63, 2v69, 2v6a - CrRBCO (mutant)
1mlv - pRBCO LSMT – pea
2cxe, 2cwx – PhRBCO - Pyrococcus horikoshii
1uzd – CrRBCO/spRBCO
1geh – TkRBCO – Thermococcus kodakaraensis
1iwa - GpRBCO – Galdieria partita
1tel – RBCO large subunit – Chlorobium tepidum
RuBisCO complex with inhibitor 2-CABP
3kdn, 3a12 – TkRBCO III + 2-CABP
3kdo, 3a13 - TkRBCO III (mutant) + 2-CABP
1ir2 - CrRBCO + 2-CABP
1upm, 1upp, 1rbo, 3ruc, 8ruc - spRBCO + 2-CABP + Cation
1ir1 - spRBCO + 2-CABP + CO2 + Mg
1wdd – RBCO + 2-CABP – rice
1bwv - GpRBCO + 2-CABP
RuBisCO complex with product
1aa1 – spRBCO + phosphoglycerate
1rus - RrRBCO + phosphoglycerate
RuBisCO complex with substrate
1rcx, 1rxo – spRBCO + ribulose-1,5-bisphosphate
RuBisCO complexes
2h21 – pRBCO LSMT + AdoMet
2h23 - pRBCO LSMT + AdoHcy
2h2e, 1ozv, 1p0y - pRBCO LSMT + AdoMet + lysine
2h2j - pRBCO LSMT + sinefungin
2d69 – PhRBCO + sulfate
1rco - spRBCO + xylulose-diol-1,5-bisphosphate
2rus - RrRBCO + CO2 + Mg
1ej7 – RBCO + phosphate - tobacco
Notes and Literarture References
- ↑ Image of the Calvin cyclewas obtained from Wikipedia
- ↑ R. Ellis The most abundant protein in the world doi:10.1016/0968-0004(79)90212-3
- ↑ Imagewas obtained from Wikipedia
- ↑ Bowes G, Ogren WL, Hageman RH. Phosphoglycolate production catalyzed by ribulose diphosphate carboxylase. Biochem Biophys Res Commun. 1971 Nov 5;45(3):716-22. PMID:4331471
- ↑ Imagewas obtained from Wikipedia
- ↑ 6.0 6.1 P. Schopfer und A. Brennicke: Pflanzenphysiologie. 6. Auflage. Elsevier, 2005,
- ↑ Yoshida S, Atomi H, Imanaka T. Engineering of a type III rubisco from a hyperthermophilic archaeon in order to enhance catalytic performance in mesophilic host cells. Appl Environ Microbiol. 2007 Oct;73(19):6254-61. Epub 2007 Aug 3. PMID:17675435 doi:10.1128/AEM.00044-07
- ↑ 8.0 8.1 Andersson I. Large structures at high resolution: the 1.6 A crystal structure of spinach ribulose-1,5-bisphosphate carboxylase/oxygenase complexed with 2-carboxyarabinitol bisphosphate. J Mol Biol. 1996 May 31;259(1):160-74. PMID:8648644 doi:10.1006/jmbi.1996.0310
- ↑ 9.0 9.1 9.2 9.3 9.4 9.5 9.6 9.7 9.8 9.9 Whitney SM, Houtz RL, Alonso H. Advancing our understanding and capacity to engineer nature's CO2-sequestering enzyme, Rubisco. Plant Physiol. 2011 Jan;155(1):27-35. Epub 2010 Oct 25. PMID:20974895 doi:10.1104/pp.110.164814
- ↑ Cleland WW, Andrews TJ, Gutteridge S, Hartman FC, Lorimer GH. Mechanism of Rubisco: The Carbamate as General Base. Chem Rev. 1998 Apr 2;98(2):549-562. PMID:11848907
- ↑ Imagefrom the Molecule of the Month at RCSB PDB
- ↑ doiF. C. Hartman, C. D. Stringer, Sylvia Milanez and Eva H. Lee The Active Site of Rubisco doi: 10.1098/rstb.1986.0045 Phil. Trans. R. Soc. Lond. B 14 October 1986 vol. 313 no. 1162 379-395
Additional Literature and Resources
Jeremy M Berg, John L Tymoczko, and Lubert Stryer et al.: Biochemistry 6th edition W. H. Freeman and Company 2007
Proteopedia Page Contributors and Editors
Nicolai Rügen and Jakob Raphael Käppler