This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
User:Karl E. Zahn/Sandbox RB69 1/palm-domain
From Proteopedia
|
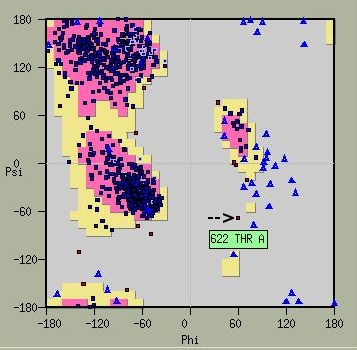
The two-metal ion mechanism employed during the DNA pol catalytic cycle relies on coordination of divalent cations by conserved aspartate motifs in the [1][2]. The palm domain of RB69 DNA pol (residues 383-468, 573-729) contains such (LDFASLYPSI and DTDS) that are particular to family B DNA pols. Interestingly, the residue T622 in this sequence must obtain a high energy conformation in order to correctly orient the D621 and D623. This property is evident in Ramachandran plots of RB69 DNA pol where T622 is an obvious out-layer. L411 and D412 complete the coordination sphere of metal ion B.
A common strategy for crystallizing DNA polymerases in complex with both DNA and an incoming nucleoside triphosphate utilizes a dideoxy-terminated primer strand in the DNA molecule. Since the 3'-OH is absent, the reaction can not proceed following binding and alignment of the substrates. However, since the 3'-OH is required to complete contacts to the coordination sphere of metal ion A, complexes with chain terminated primers often illustrate perturbation or absence of this ion[3]. Metal B is absent in the structure 2OZS.
References
- ↑ Steitz TA, Steitz JA. A general two-metal-ion mechanism for catalytic RNA. Proc Natl Acad Sci U S A. 1993 Jul 15;90(14):6498-502. PMID:8341661
- ↑ Doublie S, Tabor S, Long AM, Richardson CC, Ellenberger T. Crystal structure of a bacteriophage T7 DNA replication complex at 2.2 A resolution. Nature. 1998 Jan 15;391(6664):251-8. PMID:9440688 doi:http://dx.doi.org/10.1038/34593
- ↑ Batra VK, Beard WA, Shock DD, Krahn JM, Pedersen LC, Wilson SH. Magnesium-induced assembly of a complete DNA polymerase catalytic complex. Structure. 2006 Apr;14(4):757-66. PMID:16615916 doi:http://dx.doi.org/10.1016/j.str.2006.01.011