Function
RhoA (Ras homology gene family member A) is a protein of the small GTPase family. It can be in two conformations, and therefore active, or and consequently inactive. Three factors regulate these two states [1]:
1. GEF (Guanine nucleotide exchange factors): promotes the exchange of GDP for GTP, activating RhoA. 6bc0
2. GAP (GTPase activating proteins): accelerates the hydrolysis of GTP, inhibiting RhoA. 6pxb
3. GDI (Guanine nucleotide dissociation inhibitor): translocates the membrane GTPase, sequestering it to the cytosol, also inhibiting RhoA.
This protein is an essential molecular switch for activities associated with the cytoskeleton and the immune system. In relation to the cytoskeleton, it is a regulator of cell migration, contributing to cell retraction through the ROCK (Rho-associated protein kinase) and LIMK (LIM kinase) pathway, which leads to contraction of actomyosin II and actin polymerization. In addition, it is relevant for the assembly of occlusive junctions that seal the epithelium in selective permeability barriers, such as in the intestine. When it comes to the immune system, it is essential for the presentation of antigens and formation of immune synapses between the dendritic cell and the T lymphocyte. Not only that, but it also contributes to the recruitment and phagocytosis activity of neutrophils, macrophages and dendritic cells [2].
Disease
Mutations in RHOA have been linked to a predisposition to autoimmune diseases and cancer progression [3]. Additionally, RhoA signaling is possibly involved in the pathogenesis of neurodegenerative diseases, including Parkinson’s disease (PD), Alzheimer’s disease (AD), Huntington’s disease (HD), and amyotrophic lateral sclerosis (ALS) [4]. This may be related to the implication of Rho GTPases in brain development since these neurodegenerative diseases present an abnormal accumulation of misfolded peptides. One of these proteins could be RhoA.
In addition, different bacteria use a pathogenic strategy of inactivating RhoA through their toxins, which make post-translational modifications in the switch I region. The bacteria and their respective toxins are:
- Vibrio parahaemolyticus: VopS (adenylation of Thr 37)
- Histophilus somni: IbpA (adenylation of Try 34)
- Clostridium botulinum: C3 (ADP-ribosylation of Asn 41)
- Clostridium difficile: TcdB/A (glycosylation of Thr 37)
- Burkholderia cenocepacia: deamidation of Asn 41 [5]
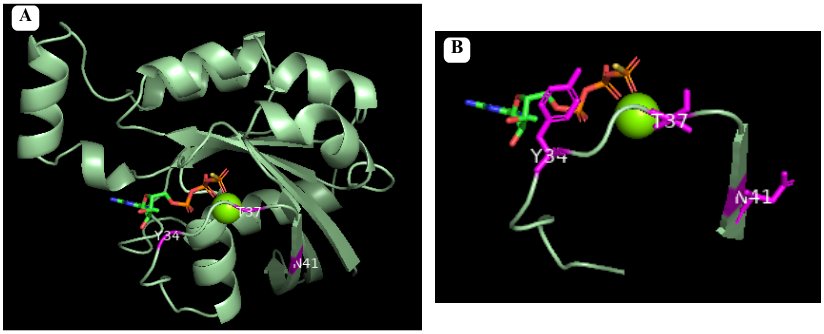
Pymol representation of RhoA with residues comumly modificated in disises(in magenta). Figure A shows the whole protein and Figure B shows the switch 1 region with the modified residues in stick representation.
Structure
RhoA is a monomeric protein comprise of 193 amino acids and its is characterized by the presence of a beta sheet (in yellow) surrounded by alfa helix (in pink) and 310 helix (in purple) connected by loops [6]. Within the RhoA protein, distinct regions can be identified, each with specific functions:
: This domain is responsible for binding and hydrolyzing . Multiple parts of the protein are involved in the activity of this region , Mg ion is also an important element, without which the affinity decreases more than 500-fold [7].
Switch I and Switch II: These are two regions within the GTPase domain that undergo conformational changes upon GTP binding. In the following green links Switch I (in dark blue) and Switch II (in blue) can be seen in or . Their conformations dictate the ability of RhoA to interact with downstream effector proteins.
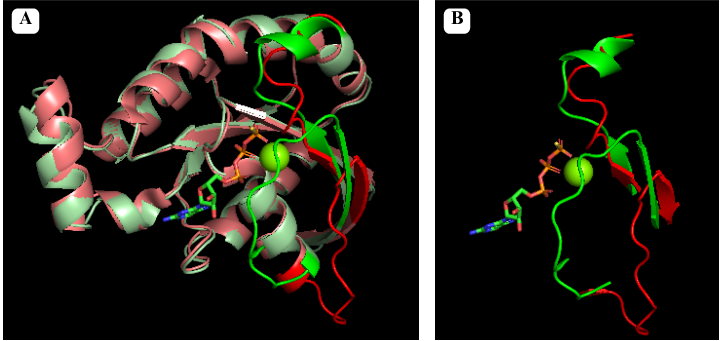
Pymol alignment of GTP bound RhoA in green and GDP bound RhoA in red. Figure A shows the full alignment with Switch I and II represented with more saturated colors. Figure B shows just Switch I and II
: This is a unique sequence insertion found within the GTPase domain of RhoA. It plays a role in the regulation and interaction of RhoA with other proteins.
: The C-terminal region (in orange) of RhoA undergoes prenylation, a post-translational modification where a prenyl lipid group (such as farnesyl or geranylgeranyl) is attached. Prenylation allows RhoA to anchor to cell membranes, facilitating its localization and interaction with membrane-associated proteins.
Post-Translational Modifications
Prenylation: The activation of Rho GTPase requires membrane binding, which is necessary for the interaction with membranous GEFs. The membrane association requires C-terminal prenylation, which involves the addition of a geranylgeranyl (20-carbon chain) to Cys190 in the CAAX motif.
Phosphorylation: Can alter the subcellular localization of RhoA when occurs close to C-terminal lipid modifications. On the other hand, phosphorylation of the G-domain affects GTP/GDP cycling and the interaction with effector proteins.
Oxidation: RhoA can be oxidized on Cys16 and Cys20 (G1 domain), generating a disulfide bond that prevents guanine binding and GEF association, inactivating RhoA. However, if Tyr42 is phosphorylated, serving as a binding site for GEF, oxidation on Cys16/20 reduces the affinity of RhoA for GDI and increases the association with GEF, leading to RhoA activation.
Nitration: Nitration on RhoA's Tyr34 (switch I region) introduces a negative charge that modifies the protein structure and leads to a faster GDP release and GTP reload, increasing RhoA activity.
Adenylation: Adenylation on Tyr34 (switch I region) leads to RhoA inhibition.
Ubiquitination: target the protein for degradation by the proteasome. RhoA is ubiquitylated by E3 ubiquitin protein ligase complexes, that ubiquitinate either active RhoA on Lys6 and Lys7, inactive RhoA, or both states on Lys135.[8]
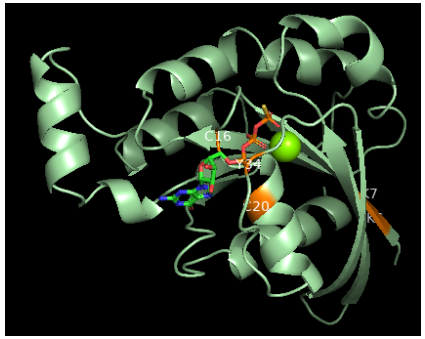
Pymol representation of RhoA with pos-translational altered residues colored as orange



