This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
User:Nathan Roy
From Proteopedia
Contents |
HIV-1 Gag
The HIV-1 Gag protein is the major structural protein required for virus assembly. It is synthesized as a polyprotein in the cytosol of an infected cell, and contains four functional segments; MA, CA (NTD and CTD), NC, and p6. The NC region is flanked by two "spacer" segments, denoted SP1 and SP2. The polyprotein is all alpha helical, except the NC region, which is composed of two RNA interacting zinc knuckle domains. Gag is often referred to as an "assembly machine" because expression of Gag alone is sufficient to produce budding virus-like particles (VLP's), due to multimerization of roughly 2000 Gag molecules per virion. Here, we will take a closer look at the MA, CA, and NC domains, and how the structural components of these domains aid in the assembly of virus particles. Viral particles can be classified as immature (pre-budding and non-infectious), and mature (post-budding and infectious), and this exchange is mediated by the HIV-1 protease(LINK). Upon viral budding, Gag is cleaved by the HIV-1 protease at multiple sites, thus possibly changing many of the structural interactions that make up the "immature" particle. For simplicity, we will only be discussing the immature formation of Gag on the plasma membrane of infected cells, as it coordinates organized viral budding. Also, it is thought that Gag forms a hexamer structure (deduced from electron microscopy studies) upon virus assembly, but because of the difficulties encountered by attempting to crystallize a multimeric structure, the exact formation of the hexamer is still up for debate. Current models predict that the MA and CA domains interact to form trimers, and these trimers conglomerate and display hexagonal symmetry.
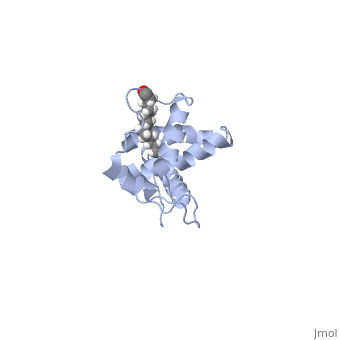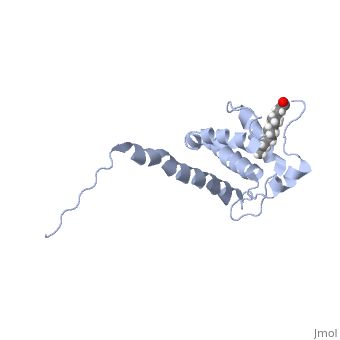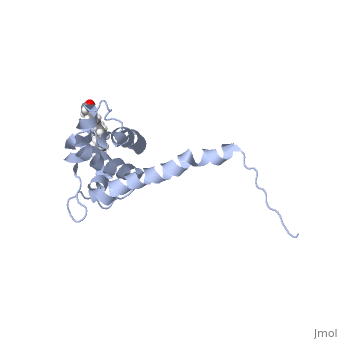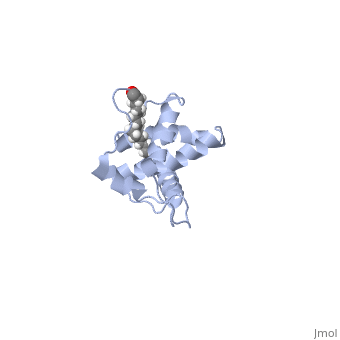
Matrix (MA)
|
|
Capsid (CA)
|
|
Note the CA NTD was not discussed here. Through biochemical studies, the NTD domain has been implicated in viral capsid assembly, but reliable structural data giving biological insights has been elusive.
Nucleocapsid (NC)
|
As we have seen, Gag is responsible for correct targeting of viral assembly to discreet sites on the plasma membrane, and viral capsid structure assembly into organized virus particles. However, Gag is also responsible for packaging of the viral RNA into budding virions, and this function in executed by the NC domain. The 5' LTR of HIV-1 genomic RNA contains a recognition element called the psi element. All retroviruses contain some type of psi element in order to get specific packaging of viral genomic RNA within the budding particles, and in the case of HIV-1, the psi element is 120 bases, and contains 4 stem-loop structures. Although the psi element can be quite variable, the SL3 loop is highly conserved within HIV-1 strains. Figure 5 shows the NC domain of HIV-1 NL4-3 in complex with the SL3 loop of the viral psi element. There are two zinc knuckle domains, with the zinc ion held by three Cys residues and a His(). Notice that guanine210 and guanine212 of the RNA interact with the F2 and F1 CCHC zinc knuckles respectively. In the F1 knuckle, G9 fits into a hydrophobic cleft formed by Val13, Phe16, Ile24, and Ala 25. G9 satisfies Watson-Crick hydrogen bonding by interacting with the NH groups from Phe16 and Ala25(), and also the CO group of Lys14 (). G7 interacts much the same with the F2 knuckle by hydrogen bonding with the NH groups of Trp37 and Met46, while also hydrogen bonding with the CO group of Gly35. The adenine211 nucleotide forms a hydrogen bond with a highly conserved Arg32 residue(). Also, residues 3 thru 10 form a 3.10 helix, which nestles into the RNA major groove(). These interactions allow viral RNA to be specifically packaged into virions. Also of note, it has been shown that NC binding of viral RNA increases the ability of Gag to multimerize, thus providing another mechanism to couple functional virion assembly to Gag multimerization and viral budding.
Reference
- De Guzman RN, Wu ZR, Stalling CC, Pappalardo L, Borer PN, Summers MF. Structure of the HIV-1 nucleocapsid protein bound to the SL3 psi-RNA recognition element. Science. 1998 Jan 16;279(5349):384-8. PMID:9430589
- Ivanov D, Tsodikov OV, Kasanov J, Ellenberger T, Wagner G, Collins T. Domain-swapped dimerization of the HIV-1 capsid C-terminal domain. Proc Natl Acad Sci U S A. 2007 Mar 13;104(11):4353-8. Epub 2007 Mar 5. PMID:17360528
- Gamble TR, Yoo S, Vajdos FF, von Schwedler UK, Worthylake DK, Wang H, McCutcheon JP, Sundquist WI, Hill CP. Structure of the carboxyl-terminal dimerization domain of the HIV-1 capsid protein. Science. 1997 Oct 31;278(5339):849-53. PMID:9346481
- Ganser-Pornillos BK, Yeager M, Sundquist WI. The structural biology of HIV assembly. Curr Opin Struct Biol. 2008 Apr;18(2):203-17. Epub 2008 Apr 9. PMID:18406133 doi:10.1016/j.sbi.2008.02.001
- Saad JS, Miller J, Tai J, Kim A, Ghanam RH, Summers MF. Structural basis for targeting HIV-1 Gag proteins to the plasma membrane for virus assembly. Proc Natl Acad Sci U S A. 2006 Jul 25;103(30):11364-9. Epub 2006 Jul 13. PMID:16840558
- Alfadhli A, Barklis RL, Barklis E. HIV-1 matrix organizes as a hexamer of trimers on membranes containing phosphatidylinositol-(4,5)-bisphosphate. Virology. 2009 May 10;387(2):466-72. Epub 2009 Mar 27. PMID:19327811 doi:10.1016/j.virol.2009.02.048
- Jouvenet N, Bieniasz PD, Simon SM. Imaging the biogenesis of individual HIV-1 virions in live cells. Nature. 2008 Jul 10;454(7201):236-40. Epub 2008 May 25. PMID:18500329 doi:10.1038/nature06998