This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
User:Tilman Schirmer/Sandbox 125
From Proteopedia
β-hairpin
|
Common motif of arrangement of two antiparallel β-strands connected by a regular turn.
(atom colors)
use better example! Note: some side-chains have multiple conformations.
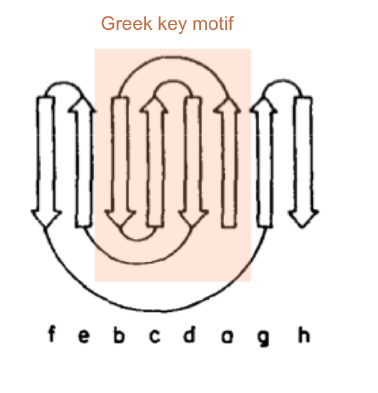
Greek key topology
|
The prealbumin is a β-sandwich composed of an antiparallel and a mixed β-sheet. It forms a with the mixed β- sheets associated to a large transmolecular sheet.
Topology diagram of the monomer:
A consists of 4 antiparallel β-strands in the order bcda, i.e. the the 1st and 4th strand are adjacent and the 2nd and 3rd strand form a β-hairpin.