This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Sandbox Reserved 347
From Proteopedia
| Line 6: | Line 6: | ||
{{STRUCTURE_2iko | PDB=2iko | SCENE=Sandbox_Reserved_347/Close_up/1 }} | {{STRUCTURE_2iko | PDB=2iko | SCENE=Sandbox_Reserved_347/Close_up/1 }} | ||
=Introduction= | =Introduction= | ||
| - | <scene name='Sandbox_Reserved_347/Close_up/1'>Renin</scene> (pronounced /ˈriːnɨn/ REE-nin) is also known as angiotensinogenase, a monospecific enzyme that participates in the body's renin-angiotensin system (RAS). Renin is responsible for catalyzing the rate-limiting step in the synthesis of angiotensin II. Once renin and pro-renin bind to the pro-renin receptor, there is an increased enzymatic activity and additional physiological effects. <ref name= | + | <scene name='Sandbox_Reserved_347/Close_up/1'>Renin</scene> (pronounced /ˈriːnɨn/ REE-nin) is also known as angiotensinogenase, a monospecific enzyme that participates in the body's renin-angiotensin system (RAS). Renin is responsible for catalyzing the rate-limiting step in the synthesis of angiotensin II. Once renin and pro-renin bind to the pro-renin receptor, there is an increased enzymatic activity and additional physiological effects. <ref name="hypertension">doi:10.1016/j.jacc.2007.10.027 |
</ref> | </ref> | ||
==Structure== | ==Structure== | ||
| - | Renin belongs in the family called aspartic proteases because they use an aspartate residue for catalysis of their peptide substrate.<ref name= | + | Renin belongs in the family called aspartic proteases because they use an aspartate residue for catalysis of their peptide substrate.<ref name="hypertension"/> |
==Biochemistry== | ==Biochemistry== | ||
Revision as of 04:05, 4 April 2011
| This Sandbox is Reserved from January 10, 2010, through April 10, 2011 for use in BCMB 307-Proteins course taught by Andrea Gorrell at the University of Northern British Columbia, Prince George, BC, Canada. |
To get started:
More help: Help:Editing |
| |||||||||
| 2iko, resolution 1.90Å () | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Ligands: | |||||||||
| Gene: | REN (Homo sapiens) | ||||||||
| Activity: | Renin, with EC number 3.4.23.15 | ||||||||
| |||||||||
| |||||||||
| Resources: | FirstGlance, OCA, RCSB, PDBsum | ||||||||
| Coordinates: | save as pdb, mmCIF, xml | ||||||||
Contents |
Introduction
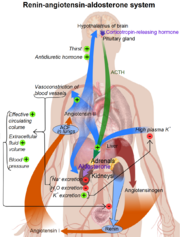
(pronounced /ˈriːnɨn/ REE-nin) is also known as angiotensinogenase, a monospecific enzyme that participates in the body's renin-angiotensin system (RAS). Renin is responsible for catalyzing the rate-limiting step in the synthesis of angiotensin II. Once renin and pro-renin bind to the pro-renin receptor, there is an increased enzymatic activity and additional physiological effects. [1]
Structure
Renin belongs in the family called aspartic proteases because they use an aspartate residue for catalysis of their peptide substrate.[1]
Biochemistry
Renin is an aspartyl protease. [2]
|
Function
Renin plays a key role in the Renin-Angiotension sysmtem (RAS). This system is responsible for the control of blood pressure and salt balances in mammals.
|