This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Amyloid precursor protein
From Proteopedia
| Line 1: | Line 1: | ||
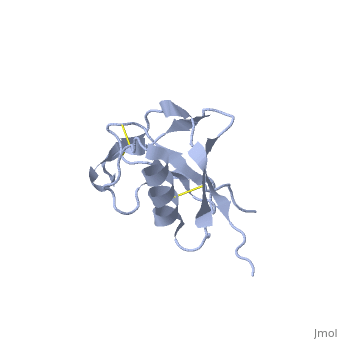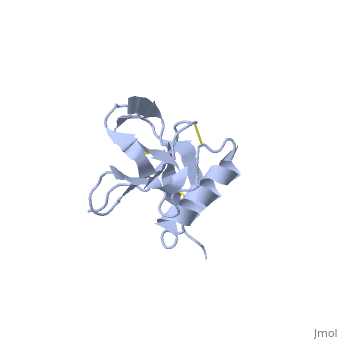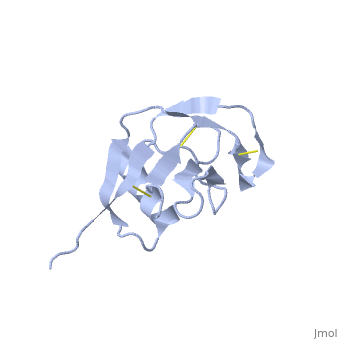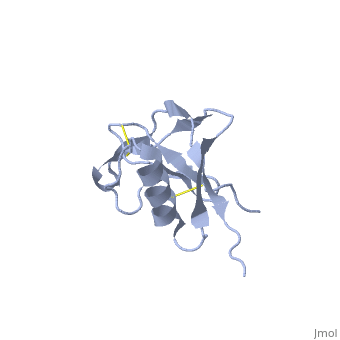
{{STRUCTURE_1mwp| PDB=1mwp | SIZE=400| SCENE= |right| CAPTION=Human amyloid precursor protein heparin-binding domain [[1mwp]] }} | {{STRUCTURE_1mwp| PDB=1mwp | SIZE=400| SCENE= |right| CAPTION=Human amyloid precursor protein heparin-binding domain [[1mwp]] }} | ||
| - | '''Amyloid precursor protein''' (APP) is thought to regulate transcription. For detailed discussion see [[Human APP]] | + | '''Amyloid precursor protein''' (APP) is thought to regulate transcription. APP is cleaved by β-secretase and the resulting N terminal peptide which is ca. 40 amino acid long is called hAPP β-peptide. The aggregation of the hAPP β-peptide is the cause of Alzheimer’s Disease. For detailed discussion see<br /> |
| + | * [[Human APP]]<br /> | ||
| + | * [[Human APP Intracellular Domain Complex with Fe65-PTB2]]<br /> | ||
| + | * [[Amyloid beta]]. | ||
__NOTOC__ | __NOTOC__ | ||
| Line 11: | Line 14: | ||
[[3nyj]], [[3nyl]], [[3umh]], [[3umi]], [[3umk]] – hAPP E2 domain – human<BR /> | [[3nyj]], [[3nyl]], [[3umh]], [[3umi]], [[3umk]] – hAPP E2 domain – human<BR /> | ||
[[3ktm]] – hAPP residues 18-190<BR /> | [[3ktm]] – hAPP residues 18-190<BR /> | ||
| - | [[1z0q]], [[2beg]] – hAPP β-peptide – NMR<BR /> | ||
[[1ze7]], [[2bp4]] – hAPP zinc-binding domain – NMR<BR /> | [[1ze7]], [[2bp4]] – hAPP zinc-binding domain – NMR<BR /> | ||
[[2fjz]], [[2fma]] - hAPP residues 133-189<BR /> | [[2fjz]], [[2fma]] - hAPP residues 133-189<BR /> | ||
| Line 19: | Line 21: | ||
[[1qyt]], [[1qwp]], [[1qxc]] - hAPP residues 25-35 – NMR<BR /> | [[1qyt]], [[1qwp]], [[1qxc]] - hAPP residues 25-35 – NMR<BR /> | ||
[[1mwp]] - hAPP heparin-binding domain<br /> | [[1mwp]] - hAPP heparin-binding domain<br /> | ||
| + | |||
| + | ===hAPP β-peptide=== | ||
| + | |||
| + | [[1z0q]], [[2beg]] – hAPP β-peptide residues 1-42 – NMR<br /> | ||
| + | [[1amb]], [[1amc]] – hAPP β-peptide residues 1-28 – NMR<br /> | ||
| + | [[1qcm]], [[1qwp]], [[1qxc]], [[1qyt]] – hAPP β-peptide residues 25-35 – NMR<br /> | ||
| + | [[1ba4]], [[1ba6]], [[2lnq]] – hAPP β-peptide residues 1-40 – NMR<br /> | ||
| + | [[1bjb]], [[1bjc]] – hAPP β-peptide residues 1-28 (mutant) – NMR<br /> | ||
| + | [[1nmj]] – APP β-peptide residues 1-28 – rat – NMR<br /> | ||
| + | [[3bae]] – hAPP β-peptide residues 1-28 + antibody<br /> | ||
| + | [[3bkj]] – hAPP β-peptide residues 1-16 + antibody<br /> | ||
| + | [[2roz]] – hAPP β-peptide residues 1-32 + amyloid β A4 precursor protein - NMR<br /> | ||
| + | [[2wk3]] – hAPP β-peptide residues 1-42 + insulin-degrading enzyme<br /> | ||
===Amyloid precursor protein binary complex=== | ===Amyloid precursor protein binary complex=== | ||
Revision as of 09:23, 9 April 2013
| |||||||||
| Human amyloid precursor protein heparin-binding domain 1mwp | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| |||||||||
| |||||||||
| Resources: | FirstGlance, OCA, RCSB, PDBsum | ||||||||
| Coordinates: | save as pdb, mmCIF, xml | ||||||||
Amyloid precursor protein (APP) is thought to regulate transcription. APP is cleaved by β-secretase and the resulting N terminal peptide which is ca. 40 amino acid long is called hAPP β-peptide. The aggregation of the hAPP β-peptide is the cause of Alzheimer’s Disease. For detailed discussion see
3D structures of amyloid precursor protein
Updated on 09-April-2013
3nyj, 3nyl, 3umh, 3umi, 3umk – hAPP E2 domain – human
3ktm – hAPP residues 18-190
1ze7, 2bp4 – hAPP zinc-binding domain – NMR
2fjz, 2fma - hAPP residues 133-189
1owt - hAPP residues 133-189 - NMR
1rw6 - hAPP residues 346-551
1tkn - hAPP residues 460-569 - NMR
1qyt, 1qwp, 1qxc - hAPP residues 25-35 – NMR
1mwp - hAPP heparin-binding domain
hAPP β-peptide
1z0q, 2beg – hAPP β-peptide residues 1-42 – NMR
1amb, 1amc – hAPP β-peptide residues 1-28 – NMR
1qcm, 1qwp, 1qxc, 1qyt – hAPP β-peptide residues 25-35 – NMR
1ba4, 1ba6, 2lnq – hAPP β-peptide residues 1-40 – NMR
1bjb, 1bjc – hAPP β-peptide residues 1-28 (mutant) – NMR
1nmj – APP β-peptide residues 1-28 – rat – NMR
3bae – hAPP β-peptide residues 1-28 + antibody
3bkj – hAPP β-peptide residues 1-16 + antibody
2roz – hAPP β-peptide residues 1-32 + amyloid β A4 precursor protein - NMR
2wk3 – hAPP β-peptide residues 1-42 + insulin-degrading enzyme
Amyloid precursor protein binary complex
1ze9 - hAPP zinc-binding domain + Zn – NMR
2fk1, 2fk2, 2fk3, 2fkl - hAPP residues 133-189 + Cu
3jti, 3gci – hAPP peptide + phospholipase A2
3l81 - hAPP peptide + AP-4 complex subunit μ-1
3ifl, 3ifn, 3ifo, 3ifp, 2r0w - hAPP peptide + antibody
2wk3 - hAPP residues 1-42 + insulin degrading enzyme
3dxc - hAPP residues 739-770 + Fe65-PTB2
3dxd, 3dxe - hAPP residues 739-770 (mutant) + Fe65-PTB2
2roz - hAPP peptide + Fe65 C terminal
2otk - hAPP residues 672-711 + ZAB3 affibody dimer - NMR
3l3t - hAPP residues 211-267 + mesotrypsin
3l33 - hAPP residues 290-341 + trypsin-3 (mutant)