This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Sandbox Reserved 1083
From Proteopedia
(Difference between revisions)
| Line 11: | Line 11: | ||
== Assembly within biological system == | == Assembly within biological system == | ||
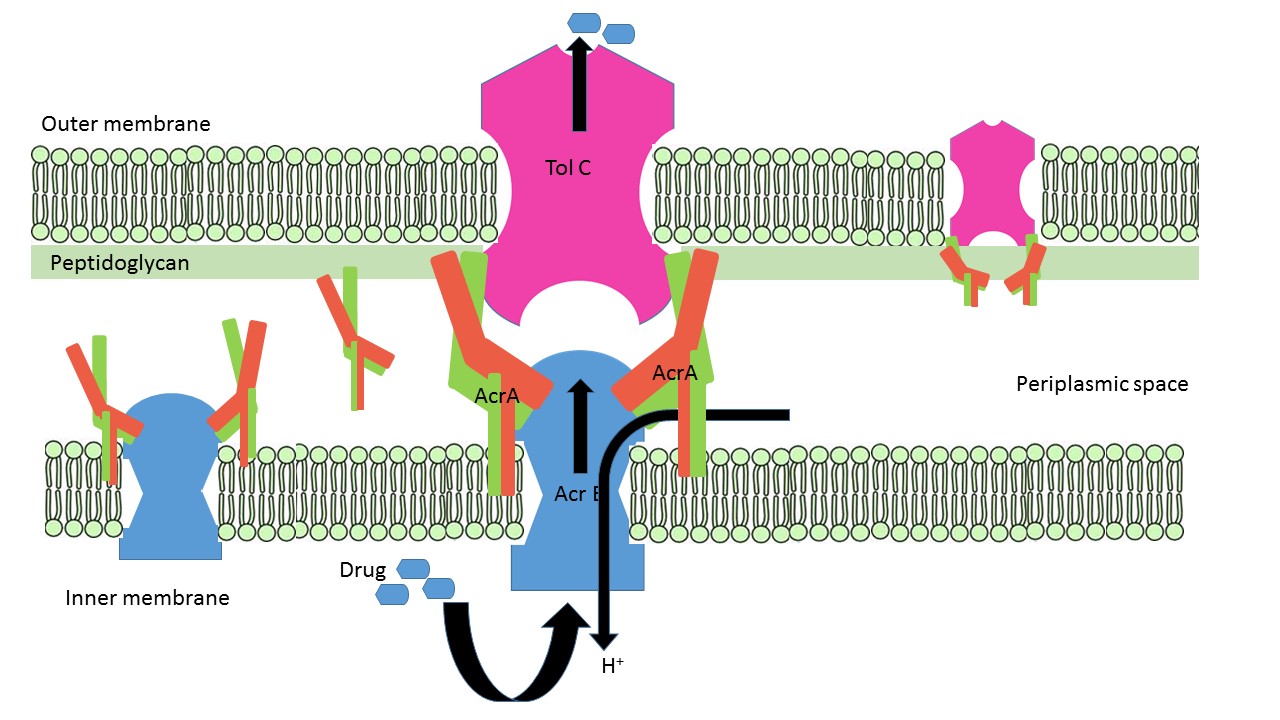
| - | AcrA is present within E. coli cells as a part of tripartite membrane associated efflux system along with [http://www.uniprot.org/uniprot/P31224 AcrB] and [http://www.uniprot.org/uniprot/P02930 TolC]. AcrA is present in the periplasmic space of cell with the proton antiporter AcrB in the inner-membrane and channel TolC in the outer membrane. It can it can remain free or form bipartite complexes with AcrB and TolC. The lipoyl and β-barrel domain of AcrA interact with AcrB, whereas the α-helical hairpin domain interact with TolC (Qiang Ge et al, 2009). ArcA remains attached to the inner membrane via lipid acylation of Cys-25. N and C termini of AcrA form two <scene name='69/699996/Membrane_proximal/1'>apposing β-stands</scene>, β1 (54-61) and β14 (292-297)lies proximal to inner membrane. A 28 flexible residues connects acylated Cys-25 with the β-barrel domain and allow the protein to reach the periplasmic top of AcrB (Yu et al 2003). A <scene name='69/699996/Short_alpha_helix/1'>short α-helix</scene> (222-230) located between β-10 (in red) and β-11 (in blue) closes off the end of the β-barrel near the C-terminus of AcrA fragment. Approximately 100 residues in the C-terminal are predicted to be important for AcrAB-TolC interaction. | + | AcrA is present within E. coli cells as a part of tripartite membrane associated efflux system along with [http://www.uniprot.org/uniprot/P31224 AcrB] and [http://www.uniprot.org/uniprot/P02930 TolC]. AcrA is present in the periplasmic space of cell with the proton antiporter AcrB in the inner-membrane and channel TolC in the outer membrane. It can it can remain free or form bipartite complexes with AcrB and TolC. The lipoyl and β-barrel domain of AcrA interact with AcrB, whereas the α-helical hairpin domain interact with TolC (Qiang Ge et al, 2009). [[Image: AcrA_AcrB_TolC_assembly.jpg]] |
| + | ArcA remains attached to the inner membrane via lipid acylation of Cys-25. N and C termini of AcrA form two <scene name='69/699996/Membrane_proximal/1'>apposing β-stands</scene>, β1 (54-61) and β14 (292-297)lies proximal to inner membrane. A 28 flexible residues connects acylated Cys-25 with the β-barrel domain and allow the protein to reach the periplasmic top of AcrB (Yu et al 2003). A <scene name='69/699996/Short_alpha_helix/1'>short α-helix</scene> (222-230) located between β-10 (in red) and β-11 (in blue) closes off the end of the β-barrel near the C-terminus of AcrA fragment. Approximately 100 residues in the C-terminal are predicted to be important for AcrAB-TolC interaction. | ||
</StructureSection> | </StructureSection> | ||
== References == | == References == | ||
<references/> | <references/> | ||
Revision as of 14:21, 22 April 2015
| This Sandbox is Reserved from 15/04/2015, through 15/06/2015 for use in the course "Protein structure, function and folding" taught by Taru Meri at the University of Helsinki. This reservation includes Sandbox Reserved 1081 through Sandbox Reserved 1090. |
To get started:
More help: Help:Editing |
AcrA
| |||||||||||