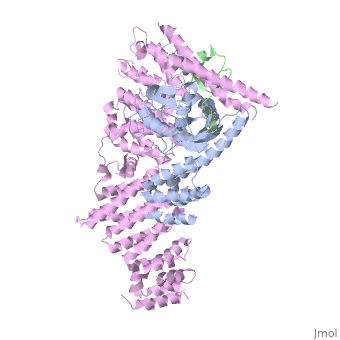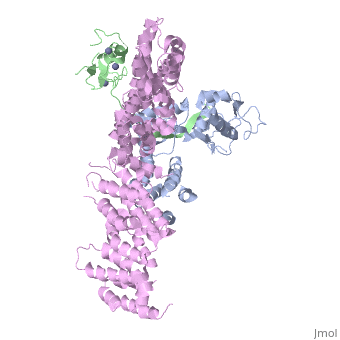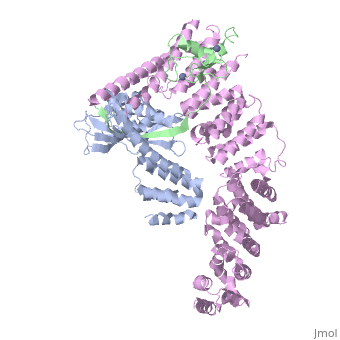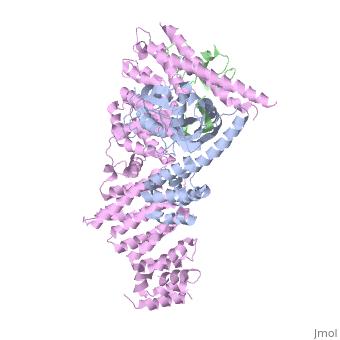This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
RING box protein
From Proteopedia
(Difference between revisions)
| Line 1: | Line 1: | ||
| - | <StructureSection load='4f52' size='340' side='right' caption='Human RING finger protein 1 (green | + | <StructureSection load='4f52' size='340' side='right' caption='Human RING finger protein 1 (green) complex with cullin-1 (grey), glomulin (magenta) and Zn+2 ions (grey) (PDB code [[4f52]])' scene=''> |
'''RING box protein''' (RBX) makes a heterodimer with cullin. The heterodimer is essential for the protein ubiquitination process. RBX1 is a component of the SCF (Skp1-cullin-F box protein) E3 ubiquitin ligase complex. RBX contains a RING-type zinc finger domain. The RING domain is 40 to 60 residues long and binds two Zn atoms. It is involved in protein-protein interaction. | '''RING box protein''' (RBX) makes a heterodimer with cullin. The heterodimer is essential for the protein ubiquitination process. RBX1 is a component of the SCF (Skp1-cullin-F box protein) E3 ubiquitin ligase complex. RBX contains a RING-type zinc finger domain. The RING domain is 40 to 60 residues long and binds two Zn atoms. It is involved in protein-protein interaction. | ||
Revision as of 09:10, 17 August 2016
| |||||||||||
3D Structures of RING box protein
Updated on 17-August-2016