Main Page
From Proteopedia
| Line 62: | Line 62: | ||
<table width='100%' style="padding: 10px; background-color: #d7d8f9; font-size: 1.5em;"><tr> | <table width='100%' style="padding: 10px; background-color: #d7d8f9; font-size: 1.5em;"><tr> | ||
<td>[[Proteopedia:About|About]]</td> | <td>[[Proteopedia:About|About]]</td> | ||
| - | <td> | + | <td>[http://proteopedia.org/cgi-bin/contact Contact]</td> |
<td>[[Proteopedia:Table of Contents|Table of Contents]]</td> | <td>[[Proteopedia:Table of Contents|Table of Contents]]</td> | ||
<td>[[Proteopedia:Structure Index|Structure Index]]</td> | <td>[[Proteopedia:Structure Index|Structure Index]]</td> | ||
Revision as of 09:09, 21 October 2018
|
ISSN 2310-6301
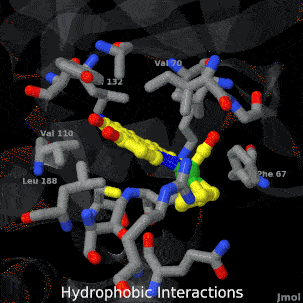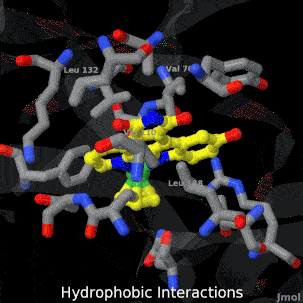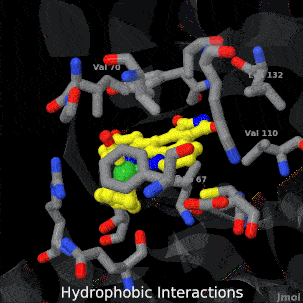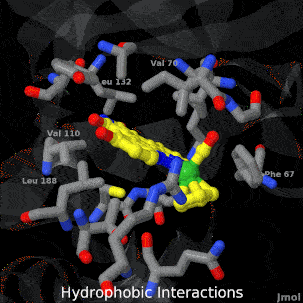
As life is more than 2D, Proteopedia helps to bridge the 3D relationships between function & structure of biomacromolecules
| |||||||||||
| Selected Pages | Art on Science | Journals | Education | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
|
|
|
|
||||||||
|
How to add content to Proteopedia Who knows ... |
Teaching Strategies Using Proteopedia |
||||||||||
| |||||||||||