|
|
| (17 intermediate revisions not shown.) |
| Line 1: |
Line 1: |
| - | [[Image:1w06.gif|left|200px]]<br /> | |
| - | <applet load="1w06" size="450" color="white" frame="true" align="right" spinBox="true" | |
| - | caption="1w06, resolution 1.65Å" /> | |
| - | '''ISOPENICILLIN N SYNTHASE AMINOADIPOYL-CYSTEINYL-ALANINE-FE NO COMPLEX'''<br /> | |
| | | | |
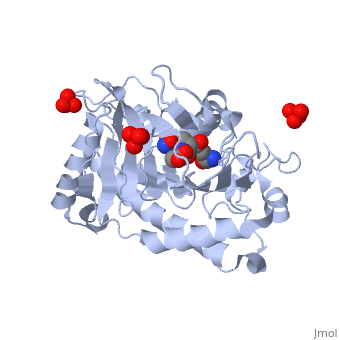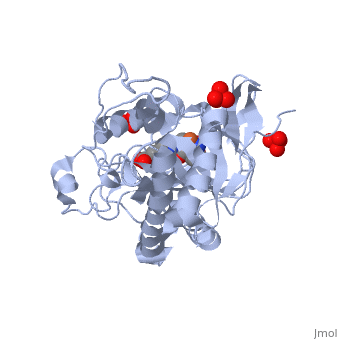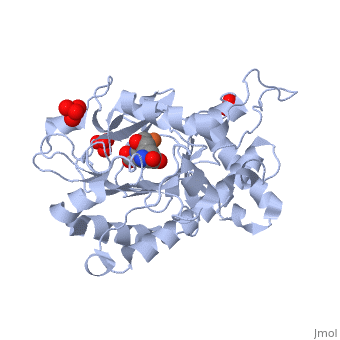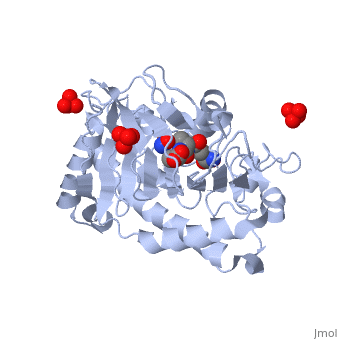
| - | ==Overview== | + | ==Isopenicillin N Synthase Aminoadipoyl-Cysteinyl-Alanine-Fe NO Complex== |
| - | Isopenicillin N synthase (IPNS), a non-heme iron(II)-dependent oxidase, catalyzes conversion of the tripeptide, delta-(l-alpha-aminoadipoyl)-l-cysteinyl-d-valine (ACV) to bicyclic, isopenicillin N (IPN), concomitant with the reduction of dioxygen to two, molecules of water. Incubation of the "truncated"substrate analogues, delta-(l-alpha-aminoadipoyl)-l-cysteinyl-glycine (ACG) and, delta-(l-alpha-aminoadipoyl)-l-cysteinyl-d-alanine (ACA) with IPNS has, previously been shown to afford acyclic products, in which the substrate, cysteinyl residue has undergone a two-electron oxidation. We report X-ray, crystal structures for the anaerobic IPNS/Fe(II)/ACG and IPNS/Fe(II)/ACA, complexes, both in the absence and presence of the dioxygen analogue, nitric oxide. The overall protein structures are very similar to those of, the corresponding IPNS/Fe(II)/ACV complexes; however, significant, differences are apparent in the vicinity of the active site iron. The, structure of the IPNS/Fe(II)/ACG complex reveals that the C-terminal, carboxylate of this substrate is oriented toward the active site iron, atom, apparently hydrogen-bonded to an additional water ligand at the, metal; this is a different binding mode to that observed in the, IPNS/Fe(II)/ACV complex. ACA binds to the metal in a manner that is, intermediate between those observed for ACV and ACG. The addition of NO to, these complexes initiates conformational changes such that both the, IPNS/Fe(II)/ACG/NO and IPNS/Fe(II)/ACA/NO structures closely resemble the, IPNS/Fe(II)/ACV/NO complex. These results further demonstrate the, feasibility of metal-centered rearrangements in catalysis by non-heme iron, enzymes and provide insight into the delicate balance between, hydrophilic-hydrophobic interactions and steric effects in the IPNS active, site. | + | <StructureSection load='1w06' size='340' side='right'caption='[[1w06]], [[Resolution|resolution]] 1.65Å' scene=''> |
| | + | == Structural highlights == |
| | + | <table><tr><td colspan='2'>[[1w06]] is a 1 chain structure with sequence from [https://en.wikipedia.org/wiki/Aspergillus_nidulans Aspergillus nidulans]. Full crystallographic information is available from [http://oca.weizmann.ac.il/oca-bin/ocashort?id=1W06 OCA]. For a <b>guided tour on the structure components</b> use [https://proteopedia.org/fgij/fg.htm?mol=1W06 FirstGlance]. <br> |
| | + | </td></tr><tr id='method'><td class="sblockLbl"><b>[[Empirical_models|Method:]]</b></td><td class="sblockDat" id="methodDat">X-ray diffraction, [[Resolution|Resolution]] 1.65Å</td></tr> |
| | + | <tr id='ligand'><td class="sblockLbl"><b>[[Ligand|Ligands:]]</b></td><td class="sblockDat" id="ligandDat"><scene name='pdbligand=FE2:FE+(II)+ION'>FE2</scene>, <scene name='pdbligand=HOA:HYDROXYAMINE'>HOA</scene>, <scene name='pdbligand=SO4:SULFATE+ION'>SO4</scene>, <scene name='pdbligand=W05:DELTA-(L-ALPHA-AMINOADIPOYL)-L-CYSTEINYL-D-ALANINE'>W05</scene></td></tr> |
| | + | <tr id='resources'><td class="sblockLbl"><b>Resources:</b></td><td class="sblockDat"><span class='plainlinks'>[https://proteopedia.org/fgij/fg.htm?mol=1w06 FirstGlance], [http://oca.weizmann.ac.il/oca-bin/ocaids?id=1w06 OCA], [https://pdbe.org/1w06 PDBe], [https://www.rcsb.org/pdb/explore.do?structureId=1w06 RCSB], [https://www.ebi.ac.uk/pdbsum/1w06 PDBsum], [https://prosat.h-its.org/prosat/prosatexe?pdbcode=1w06 ProSAT]</span></td></tr> |
| | + | </table> |
| | + | == Function == |
| | + | [https://www.uniprot.org/uniprot/IPNA_EMENI IPNA_EMENI] Isopenicillin N synthase; part of the gene cluster that mediates the biosynthesis of penicillin, the world's most important antibiotic (PubMed:3319778, PubMed:11755401). IpnA catalyzes the cyclization of the tripeptide N-[(5S)-5-amino-5-carboxypentanoyl]-L-cysteinyl-D-valine (LLD-ACV or ACV) to form isopenicillin N (IPN) that contains the beta-lactam nucleus (PubMed:3319778, PubMed:11755401, PubMed:28703303). The penicillin biosynthesis occurs via 3 enzymatic steps, the first corresponding to the production of the tripeptide N-[(5S)-5-amino-5-carboxypentanoyl]-L-cysteinyl-D-valine (LLD-ACV or ACV) by the NRPS acvA. The tripeptide ACV is then cyclized to isopenicillin N (IPN) by the isopenicillin N synthase ipnA that forms the beta-lactam nucleus. Finally, the alpha-aminoadipyl side chain is exchanged for phenylacetic acid by the isopenicillin N acyltransferase penDE to yield penicillin in the peroxisomal matrix (By similarity).[UniProtKB:P08703]<ref>PMID:11755401</ref> <ref>PMID:28703303</ref> <ref>PMID:3319778</ref> |
| | + | == Evolutionary Conservation == |
| | + | [[Image:Consurf_key_small.gif|200px|right]] |
| | + | Check<jmol> |
| | + | <jmolCheckbox> |
| | + | <scriptWhenChecked>; select protein; define ~consurf_to_do selected; consurf_initial_scene = true; script "/wiki/ConSurf/w0/1w06_consurf.spt"</scriptWhenChecked> |
| | + | <scriptWhenUnchecked>script /wiki/extensions/Proteopedia/spt/initialview01.spt</scriptWhenUnchecked> |
| | + | <text>to colour the structure by Evolutionary Conservation</text> |
| | + | </jmolCheckbox> |
| | + | </jmol>, as determined by [http://consurfdb.tau.ac.il/ ConSurfDB]. You may read the [[Conservation%2C_Evolutionary|explanation]] of the method and the full data available from [http://bental.tau.ac.il/new_ConSurfDB/main_output.php?pdb_ID=1w06 ConSurf]. |
| | + | <div style="clear:both"></div> |
| | + | <div style="background-color:#fffaf0;"> |
| | + | == Publication Abstract from PubMed == |
| | + | Isopenicillin N synthase (IPNS), a non-heme iron(II)-dependent oxidase, catalyzes conversion of the tripeptide delta-(l-alpha-aminoadipoyl)-l-cysteinyl-d-valine (ACV) to bicyclic isopenicillin N (IPN), concomitant with the reduction of dioxygen to two molecules of water. Incubation of the "truncated"substrate analogues delta-(l-alpha-aminoadipoyl)-l-cysteinyl-glycine (ACG) and delta-(l-alpha-aminoadipoyl)-l-cysteinyl-d-alanine (ACA) with IPNS has previously been shown to afford acyclic products, in which the substrate cysteinyl residue has undergone a two-electron oxidation. We report X-ray crystal structures for the anaerobic IPNS/Fe(II)/ACG and IPNS/Fe(II)/ACA complexes, both in the absence and presence of the dioxygen analogue nitric oxide. The overall protein structures are very similar to those of the corresponding IPNS/Fe(II)/ACV complexes; however, significant differences are apparent in the vicinity of the active site iron. The structure of the IPNS/Fe(II)/ACG complex reveals that the C-terminal carboxylate of this substrate is oriented toward the active site iron atom, apparently hydrogen-bonded to an additional water ligand at the metal; this is a different binding mode to that observed in the IPNS/Fe(II)/ACV complex. ACA binds to the metal in a manner that is intermediate between those observed for ACV and ACG. The addition of NO to these complexes initiates conformational changes such that both the IPNS/Fe(II)/ACG/NO and IPNS/Fe(II)/ACA/NO structures closely resemble the IPNS/Fe(II)/ACV/NO complex. These results further demonstrate the feasibility of metal-centered rearrangements in catalysis by non-heme iron enzymes and provide insight into the delicate balance between hydrophilic-hydrophobic interactions and steric effects in the IPNS active site. |
| | | | |
| - | ==About this Structure==
| + | Structural studies on the reaction of isopenicillin N synthase with the truncated substrate analogues delta-(L-alpha-aminoadipoyl)-L-cysteinyl-glycine and delta-(L-alpha-aminoadipoyl)-L-cysteinyl-D-alanine.,Long AJ, Clifton IJ, Roach PL, Baldwin JE, Rutledge PJ, Schofield CJ Biochemistry. 2005 May 3;44(17):6619-28. PMID:15850395<ref>PMID:15850395</ref> |
| - | 1W06 is a [http://en.wikipedia.org/wiki/Single_protein Single protein] structure of sequence from [http://en.wikipedia.org/wiki/Emericella_nidulans Emericella nidulans] with FE2, SO4, W05 and NO as [http://en.wikipedia.org/wiki/ligands ligands]. Active as [http://en.wikipedia.org/wiki/Isopenicillin-N_synthase Isopenicillin-N synthase], with EC number [http://www.brenda-enzymes.info/php/result_flat.php4?ecno=1.21.3.1 1.21.3.1] Structure known Active Site: AC1. Full crystallographic information is available from [http://ispc.weizmann.ac.il/oca-bin/ocashort?id=1W06 OCA].
| + | |
| | | | |
| - | ==Reference==
| + | From MEDLINE®/PubMed®, a database of the U.S. National Library of Medicine.<br> |
| - | Structural studies on the reaction of isopenicillin N synthase with the truncated substrate analogues delta-(L-alpha-aminoadipoyl)-L-cysteinyl-glycine and delta-(L-alpha-aminoadipoyl)-L-cysteinyl-D-alanine., Long AJ, Clifton IJ, Roach PL, Baldwin JE, Rutledge PJ, Schofield CJ, Biochemistry. 2005 May 3;44(17):6619-28. PMID:[http://ispc.weizmann.ac.il//pmbin/getpm?pmid=15850395 15850395]
| + | </div> |
| - | [[Category: Emericella nidulans]]
| + | <div class="pdbe-citations 1w06" style="background-color:#fffaf0;"></div> |
| - | [[Category: Isopenicillin-N synthase]]
| + | |
| - | [[Category: Single protein]]
| + | |
| - | [[Category: Clifton, I.J.]]
| + | |
| - | [[Category: Long, A.J.]]
| + | |
| - | [[Category: Rutledge, P.J.]]
| + | |
| - | [[Category: FE2]]
| + | |
| - | [[Category: NO]]
| + | |
| - | [[Category: SO4]]
| + | |
| - | [[Category: W05]]
| + | |
| - | [[Category: b-lactam antibiotic]]
| + | |
| - | [[Category: oxidoreductase]]
| + | |
| - | [[Category: oxygenase]]
| + | |
| - | [[Category: penicillin biosynthesis]]
| + | |
| | | | |
| - | ''Page seeded by [http://ispc.weizmann.ac.il/oca OCA ] on Mon Nov 5 17:21:15 2007''
| + | ==See Also== |
| | + | *[[Isopenicillin N synthase|Isopenicillin N synthase]] |
| | + | == References == |
| | + | <references/> |
| | + | __TOC__ |
| | + | </StructureSection> |
| | + | [[Category: Aspergillus nidulans]] |
| | + | [[Category: Large Structures]] |
| | + | [[Category: Clifton IJ]] |
| | + | [[Category: Long AJ]] |
| | + | [[Category: Rutledge PJ]] |
| Structural highlights
Function
IPNA_EMENI Isopenicillin N synthase; part of the gene cluster that mediates the biosynthesis of penicillin, the world's most important antibiotic (PubMed:3319778, PubMed:11755401). IpnA catalyzes the cyclization of the tripeptide N-[(5S)-5-amino-5-carboxypentanoyl]-L-cysteinyl-D-valine (LLD-ACV or ACV) to form isopenicillin N (IPN) that contains the beta-lactam nucleus (PubMed:3319778, PubMed:11755401, PubMed:28703303). The penicillin biosynthesis occurs via 3 enzymatic steps, the first corresponding to the production of the tripeptide N-[(5S)-5-amino-5-carboxypentanoyl]-L-cysteinyl-D-valine (LLD-ACV or ACV) by the NRPS acvA. The tripeptide ACV is then cyclized to isopenicillin N (IPN) by the isopenicillin N synthase ipnA that forms the beta-lactam nucleus. Finally, the alpha-aminoadipyl side chain is exchanged for phenylacetic acid by the isopenicillin N acyltransferase penDE to yield penicillin in the peroxisomal matrix (By similarity).[UniProtKB:P08703][1] [2] [3]
Evolutionary Conservation
Check, as determined by ConSurfDB. You may read the explanation of the method and the full data available from ConSurf.
Publication Abstract from PubMed
Isopenicillin N synthase (IPNS), a non-heme iron(II)-dependent oxidase, catalyzes conversion of the tripeptide delta-(l-alpha-aminoadipoyl)-l-cysteinyl-d-valine (ACV) to bicyclic isopenicillin N (IPN), concomitant with the reduction of dioxygen to two molecules of water. Incubation of the "truncated"substrate analogues delta-(l-alpha-aminoadipoyl)-l-cysteinyl-glycine (ACG) and delta-(l-alpha-aminoadipoyl)-l-cysteinyl-d-alanine (ACA) with IPNS has previously been shown to afford acyclic products, in which the substrate cysteinyl residue has undergone a two-electron oxidation. We report X-ray crystal structures for the anaerobic IPNS/Fe(II)/ACG and IPNS/Fe(II)/ACA complexes, both in the absence and presence of the dioxygen analogue nitric oxide. The overall protein structures are very similar to those of the corresponding IPNS/Fe(II)/ACV complexes; however, significant differences are apparent in the vicinity of the active site iron. The structure of the IPNS/Fe(II)/ACG complex reveals that the C-terminal carboxylate of this substrate is oriented toward the active site iron atom, apparently hydrogen-bonded to an additional water ligand at the metal; this is a different binding mode to that observed in the IPNS/Fe(II)/ACV complex. ACA binds to the metal in a manner that is intermediate between those observed for ACV and ACG. The addition of NO to these complexes initiates conformational changes such that both the IPNS/Fe(II)/ACG/NO and IPNS/Fe(II)/ACA/NO structures closely resemble the IPNS/Fe(II)/ACV/NO complex. These results further demonstrate the feasibility of metal-centered rearrangements in catalysis by non-heme iron enzymes and provide insight into the delicate balance between hydrophilic-hydrophobic interactions and steric effects in the IPNS active site.
Structural studies on the reaction of isopenicillin N synthase with the truncated substrate analogues delta-(L-alpha-aminoadipoyl)-L-cysteinyl-glycine and delta-(L-alpha-aminoadipoyl)-L-cysteinyl-D-alanine.,Long AJ, Clifton IJ, Roach PL, Baldwin JE, Rutledge PJ, Schofield CJ Biochemistry. 2005 May 3;44(17):6619-28. PMID:15850395[4]
From MEDLINE®/PubMed®, a database of the U.S. National Library of Medicine.
See Also
References
- ↑ Ogle JM, Clifton IJ, Rutledge PJ, Elkins JM, Burzlaff NI, Adlington RM, Roach PL, Baldwin JE. Alternative oxidation by isopenicillin N synthase observed by X-ray diffraction. Chem Biol. 2001 Dec;8(12):1231-7. PMID:11755401
- ↑ McNeill LA, Brown TJN, Sami M, Clifton IJ, Burzlaff NI, Claridge TDW, Adlington RM, Baldwin JE, Rutledge PJ, Schofield CJ. Terminally Truncated Isopenicillin N Synthase Generates a Dithioester Product: Evidence for a Thioaldehyde Intermediate during Catalysis and a New Mode of Reaction for Non-Heme Iron Oxidases. Chemistry. 2017 Sep 18;23(52):12815-12824. doi: 10.1002/chem.201701592. Epub 2017, Aug 21. PMID:28703303 doi:http://dx.doi.org/10.1002/chem.201701592
- ↑ Ramon D, Carramolino L, Patino C, Sanchez F, Penalva MA. Cloning and characterization of the isopenicillin N synthetase gene mediating the formation of the beta-lactam ring in Aspergillus nidulans. Gene. 1987;57(2-3):171-81. doi: 10.1016/0378-1119(87)90120-x. PMID:3319778 doi:http://dx.doi.org/10.1016/0378-1119(87)90120-x
- ↑ Long AJ, Clifton IJ, Roach PL, Baldwin JE, Rutledge PJ, Schofield CJ. Structural studies on the reaction of isopenicillin N synthase with the truncated substrate analogues delta-(L-alpha-aminoadipoyl)-L-cysteinyl-glycine and delta-(L-alpha-aminoadipoyl)-L-cysteinyl-D-alanine. Biochemistry. 2005 May 3;44(17):6619-28. PMID:15850395 doi:http://dx.doi.org/10.1021/bi047478q
|