This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Sandbox Reserved 347
From Proteopedia
(New page: <!-- PLEASE DO NOT DELETE THIS TEMPLATE --> {{Template:Sandbox_Reserved_BCMB307}} <!-- PLEASE ADD YOUR CONTENT BELOW HERE -->) |
|||
| (31 intermediate revisions not shown.) | |||
| Line 2: | Line 2: | ||
{{Template:Sandbox_Reserved_BCMB307}} | {{Template:Sandbox_Reserved_BCMB307}} | ||
<!-- PLEASE ADD YOUR CONTENT BELOW HERE --> | <!-- PLEASE ADD YOUR CONTENT BELOW HERE --> | ||
| + | |||
| + | |||
| + | {{STRUCTURE_2iko | PDB=2iko | SCENE=Sandbox_Reserved_347/Close_up/1 }} | ||
| + | =Introduction= | ||
| + | <scene name='Sandbox_Reserved_347/Close_up/1'>Renin</scene> (pronounced /ˈriːnɨn/ REE-nin) is also known as angiotensinogenase, a monospecific enzyme that participates in the body's renin-angiotensin system (RAS). Renin is responsible for catalyzing the rate-limiting step in the synthesis of angiotensin II. Once renin and pro-renin bind to the pro-renin receptor, there is an increased enzymatic activity and additional physiological effects. <ref name="hypertension">doi:10.1016/j.jacc.2007.10.027 | ||
| + | </ref> | ||
| + | |||
| + | |||
| + | ==Structure== | ||
| + | Renin belongs in a family called aspartic proteases which use an aspartate residue for the catalysis of their peptide substrate. X-ray diffraction experiments has shown there is a striking similarity among the structures of aspartyl proteases. <ref name="3D">K Akahane, H Umeyama, S Nakagawa, I Moriguchi, S Hirose, K Iizuka, and K Murakami. "Three-dimensional structure of human renin". ''Hypertension''. 1985;7:3-12</ref> Renin consists of two homologous lobes each containing an aspartic acid. Between the lobes is the active site, which is catalyzed by the aspartic acid residues, a characteristic trait of all aspartate proteases. <ref name="hypertension"/> Renin in its full mature form has a mass of 37 kDa and contains 340 amino acids.<ref name="cloning">PMID:9556453</ref> Uniquely from other proteases, Renin has two β-carboxyl groups of Asp32 and Asp215 which protrude from each of the two lobes into the active site and has a large flap covering the <scene name='Sandbox_Reserved_347/Ligand/1'>Active site</scene>.<ref name= "Structure">PMID:2666611</ref> | ||
| + | |||
| + | |||
| + | |||
| + | <Structure load='2iko' size='500' frame='true' align='right' caption='Renin active site' scene='Sandbox_Reserved_347/Ligand/1' /> | ||
| + | |||
| + | |||
| + | |||
| + | |||
| + | ==Function== | ||
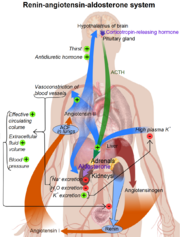
| + | [[Image:Man pic.png|thumb|left|Renin-Angiotensin-Aldosterone System]] | ||
| + | |||
| + | Renin plays a key role in the Renin-Angiotension sysmtem (RAS). It is essential in facilitating the conversion of angiotension to angiotension II, which is the active component of the RAS system.<ref name="review">DOI: 10.1146/annurev.ph.40.030178.002113</ref> This system is responsible for the regulation of blood pressure, stimulation of the secretion of aldosterone which effects the salt and water balance.<ref name="review"/> | ||
| + | *Angiotensinogen is released into the bloodstream by the liver. | ||
| + | *Likewise, Renin is secreted by the kidneys into the bloodstream where is meets with angiotensinogen. | ||
| + | *Once united, angiotensinogen form the decapeptide angiotensin (ANG) I. | ||
| + | *ANG I is then activated by Angiotensin converting enzyme (ACE) to form the octapeptide ANG II. | ||
| + | *ANG II then acts on specific receptors such as ones responsible for vasoconstriction or the release of aldosterone from the adrenal cortex. <ref name="renin review"> doi: 10.1152/physrev.00036.2005 | ||
| + | </ref> | ||
| + | |||
| + | |||
| + | |||
| + | <Structure load='2iko' size='300' frame='true' align='left' caption='Renin: Displaying the Ligand site' scene='Insert optional scene name here' /> | ||
| + | |||
| + | =References= | ||
| + | <references/> | ||
Current revision
| This Sandbox is Reserved from January 10, 2010, through April 10, 2011 for use in BCMB 307-Proteins course taught by Andrea Gorrell at the University of Northern British Columbia, Prince George, BC, Canada. |
To get started:
More help: Help:Editing |
| |||||||||
| 2iko, resolution 1.90Å () | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Ligands: | |||||||||
| Gene: | REN (Homo sapiens) | ||||||||
| Activity: | Renin, with EC number 3.4.23.15 | ||||||||
| |||||||||
| |||||||||
| Resources: | FirstGlance, OCA, RCSB, PDBsum | ||||||||
| Coordinates: | save as pdb, mmCIF, xml | ||||||||
Contents |
Introduction
(pronounced /ˈriːnɨn/ REE-nin) is also known as angiotensinogenase, a monospecific enzyme that participates in the body's renin-angiotensin system (RAS). Renin is responsible for catalyzing the rate-limiting step in the synthesis of angiotensin II. Once renin and pro-renin bind to the pro-renin receptor, there is an increased enzymatic activity and additional physiological effects. [1]
Structure
Renin belongs in a family called aspartic proteases which use an aspartate residue for the catalysis of their peptide substrate. X-ray diffraction experiments has shown there is a striking similarity among the structures of aspartyl proteases. [2] Renin consists of two homologous lobes each containing an aspartic acid. Between the lobes is the active site, which is catalyzed by the aspartic acid residues, a characteristic trait of all aspartate proteases. [1] Renin in its full mature form has a mass of 37 kDa and contains 340 amino acids.[3] Uniquely from other proteases, Renin has two β-carboxyl groups of Asp32 and Asp215 which protrude from each of the two lobes into the active site and has a large flap covering the .[4]
|
Function
Renin plays a key role in the Renin-Angiotension sysmtem (RAS). It is essential in facilitating the conversion of angiotension to angiotension II, which is the active component of the RAS system.[5] This system is responsible for the regulation of blood pressure, stimulation of the secretion of aldosterone which effects the salt and water balance.[5]
- Angiotensinogen is released into the bloodstream by the liver.
- Likewise, Renin is secreted by the kidneys into the bloodstream where is meets with angiotensinogen.
- Once united, angiotensinogen form the decapeptide angiotensin (ANG) I.
- ANG I is then activated by Angiotensin converting enzyme (ACE) to form the octapeptide ANG II.
- ANG II then acts on specific receptors such as ones responsible for vasoconstriction or the release of aldosterone from the adrenal cortex. [6]
|
References
- ↑ 1.0 1.1 Gradman AH, Kad R. Renin inhibition in hypertension. J Am Coll Cardiol. 2008 Feb 5;51(5):519-28. PMID:18237679 doi:10.1016/j.jacc.2007.10.027
- ↑ K Akahane, H Umeyama, S Nakagawa, I Moriguchi, S Hirose, K Iizuka, and K Murakami. "Three-dimensional structure of human renin". Hypertension. 1985;7:3-12
- ↑ Armstrong C. The vision of the pore. Science. 1998 Apr 3;280(5360):56-7. PMID:9556453
- ↑ Inagami T. Structure and function of renin. J Hypertens Suppl. 1989 Apr;7(2):S3-8. PMID:2666611
- ↑ 5.0 5.1 Reid IA, Morris BJ, Ganong WF. The renin-angiotensin system. Annu Rev Physiol. 1978;40:377-410. PMID:205167 doi:http://dx.doi.org/10.1146/annurev.ph.40.030178.002113
- ↑ Paul M, Poyan Mehr A, Kreutz R. Physiology of local renin-angiotensin systems. Physiol Rev. 2006 Jul;86(3):747-803. PMID:16816138 doi:10.1152/physrev.00036.2005