We apologize for Proteopedia being slow to respond. For the past two years, a new implementation of Proteopedia has been being built. Soon, it will replace this 18-year old system. All existing content will be moved to the new system at a date that will be announced here.
2uxr
From Proteopedia
(Difference between revisions)
| (7 intermediate revisions not shown.) | |||
| Line 1: | Line 1: | ||
| - | [[Image:2uxr.png|left|200px]] | ||
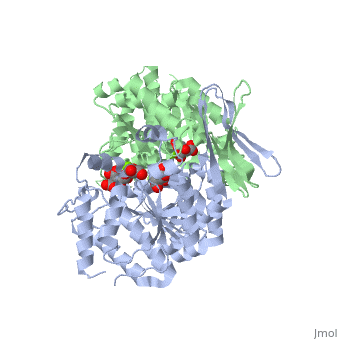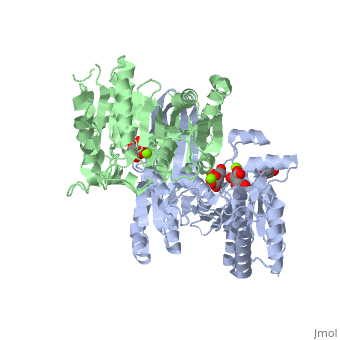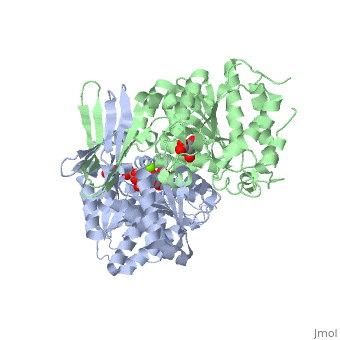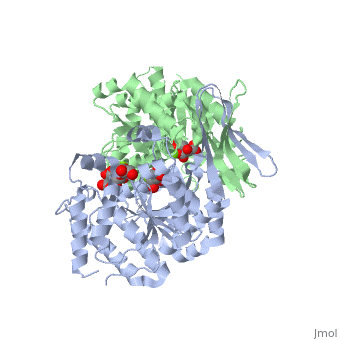
| - | + | ==Complex with isocitrate and the protein isocitrate dehydrogenase from the psychrophilic bacterium Desulfotalea psychrophila== | |
| + | <StructureSection load='2uxr' size='340' side='right'caption='[[2uxr]], [[Resolution|resolution]] 2.30Å' scene=''> | ||
| + | == Structural highlights == | ||
| + | <table><tr><td colspan='2'>[[2uxr]] is a 2 chain structure with sequence from [https://en.wikipedia.org/wiki/Desulfotalea_psychrophila Desulfotalea psychrophila]. Full crystallographic information is available from [http://oca.weizmann.ac.il/oca-bin/ocashort?id=2UXR OCA]. For a <b>guided tour on the structure components</b> use [https://proteopedia.org/fgij/fg.htm?mol=2UXR FirstGlance]. <br> | ||
| + | </td></tr><tr id='method'><td class="sblockLbl"><b>[[Empirical_models|Method:]]</b></td><td class="sblockDat" id="methodDat">X-ray diffraction, [[Resolution|Resolution]] 2.3Å</td></tr> | ||
| + | <tr id='ligand'><td class="sblockLbl"><b>[[Ligand|Ligands:]]</b></td><td class="sblockDat" id="ligandDat"><scene name='pdbligand=GOL:GLYCEROL'>GOL</scene>, <scene name='pdbligand=ICT:ISOCITRIC+ACID'>ICT</scene>, <scene name='pdbligand=MG:MAGNESIUM+ION'>MG</scene></td></tr> | ||
| + | <tr id='resources'><td class="sblockLbl"><b>Resources:</b></td><td class="sblockDat"><span class='plainlinks'>[https://proteopedia.org/fgij/fg.htm?mol=2uxr FirstGlance], [http://oca.weizmann.ac.il/oca-bin/ocaids?id=2uxr OCA], [https://pdbe.org/2uxr PDBe], [https://www.rcsb.org/pdb/explore.do?structureId=2uxr RCSB], [https://www.ebi.ac.uk/pdbsum/2uxr PDBsum], [https://prosat.h-its.org/prosat/prosatexe?pdbcode=2uxr ProSAT]</span></td></tr> | ||
| + | </table> | ||
| + | == Function == | ||
| + | [https://www.uniprot.org/uniprot/Q6AQ66_DESPS Q6AQ66_DESPS] | ||
| + | == Evolutionary Conservation == | ||
| + | [[Image:Consurf_key_small.gif|200px|right]] | ||
| + | Check<jmol> | ||
| + | <jmolCheckbox> | ||
| + | <scriptWhenChecked>; select protein; define ~consurf_to_do selected; consurf_initial_scene = true; script "/wiki/ConSurf/ux/2uxr_consurf.spt"</scriptWhenChecked> | ||
| + | <scriptWhenUnchecked>script /wiki/extensions/Proteopedia/spt/initialview01.spt</scriptWhenUnchecked> | ||
| + | <text>to colour the structure by Evolutionary Conservation</text> | ||
| + | </jmolCheckbox> | ||
| + | </jmol>, as determined by [http://consurfdb.tau.ac.il/ ConSurfDB]. You may read the [[Conservation%2C_Evolutionary|explanation]] of the method and the full data available from [http://bental.tau.ac.il/new_ConSurfDB/main_output.php?pdb_ID=2uxr ConSurf]. | ||
| + | <div style="clear:both"></div> | ||
| + | <div style="background-color:#fffaf0;"> | ||
| + | == Publication Abstract from PubMed == | ||
| + | Isocitrate dehydrogenase (IDH) has been studied extensively due to its central role in the Krebs cycle, catalyzing the oxidative NAD(P)(+)-dependent decarboxylation of isocitrate to alpha-ketoglutarate and CO(2). Here, we present the first crystal structure of IDH from a psychrophilic bacterium, Desulfotalea psychrophila (DpIDH). The structural information is combined with a detailed biochemical characterization and a comparative study with IDHs from the mesophilic bacterium Desulfitobacterium hafniense (DhIDH), porcine (PcIDH), human cytosolic (HcIDH) and the hyperthermophilic Thermotoga maritima (TmIDH). DpIDH was found to have a higher melting temperature (T(m)=66.9 degrees C) than its mesophilic homologues and a suboptimal catalytic efficiency at low temperatures. The thermodynamic activation parameters indicated a disordered active site, as seen also for the drastic increase in K(m) for isocitrate at elevated temperatures. A methionine cluster situated at the dimeric interface between the two active sites and a cluster of destabilizing charged amino acids in a region close to the active site might explain the poor isocitrate affinity. On the other hand, DpIDH was optimized for interacting with NADP(+) and the crystal structure revealed unique interactions with the cofactor. The highly acidic surface, destabilizing charged residues, fewer ion pairs and reduced size of ionic networks in DpIDH suggest a flexible global structure. However, strategic placement of ionic interactions stabilizing the N and C termini, and additional ionic interactions in the clasp domain as well as two enlarged aromatic clusters might counteract the destabilizing interactions and promote the increased thermal stability. The structure analysis of DpIDH illustrates how psychrophilic enzymes can adjust their flexibility in dynamic regions during their catalytic cycle without compromising the global stability of the protein. | ||
| - | + | Structural and functional properties of isocitrate dehydrogenase from the psychrophilic bacterium Desulfotalea psychrophila reveal a cold-active enzyme with an unusual high thermal stability.,Fedoy AE, Yang N, Martinez A, Leiros HK, Steen IH J Mol Biol. 2007 Sep 7;372(1):130-49. Epub 2007 Jun 19. PMID:17632124<ref>PMID:17632124</ref> | |
| - | + | From MEDLINE®/PubMed®, a database of the U.S. National Library of Medicine.<br> | |
| - | + | </div> | |
| - | + | <div class="pdbe-citations 2uxr" style="background-color:#fffaf0;"></div> | |
| - | + | ||
==See Also== | ==See Also== | ||
| - | *[[Ferguson sandbox 1|Ferguson sandbox 1]] | ||
*[[Isocitrate dehydrogenase|Isocitrate dehydrogenase]] | *[[Isocitrate dehydrogenase|Isocitrate dehydrogenase]] | ||
| - | + | *[[Isocitrate dehydrogenase 3D structures|Isocitrate dehydrogenase 3D structures]] | |
| - | == | + | == References == |
| - | < | + | <references/> |
| + | __TOC__ | ||
| + | </StructureSection> | ||
[[Category: Desulfotalea psychrophila]] | [[Category: Desulfotalea psychrophila]] | ||
| - | [[Category: Fedoy | + | [[Category: Large Structures]] |
| - | [[Category: Leiros | + | [[Category: Fedoy A-E]] |
| - | [[Category: Martinez | + | [[Category: Leiros H-KS]] |
| - | [[Category: Steen | + | [[Category: Martinez A]] |
| - | [[Category: Yang | + | [[Category: Steen IH]] |
| - | + | [[Category: Yang N]] | |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
Current revision
Complex with isocitrate and the protein isocitrate dehydrogenase from the psychrophilic bacterium Desulfotalea psychrophila
| |||||||||||