This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
CRISPR-Cas9
From Proteopedia
(Difference between revisions)
| (83 intermediate revisions not shown.) | |||
| Line 1: | Line 1: | ||
| - | <StructureSection load=' | + | <StructureSection load='' size='350' side='right' scene='74/742625/Cv3/13' caption='Cas9 complex with RNA (PDB code [[4zt0]])'> |
| - | + | '''Part I''' | |
| + | |||
| + | SEE ALSO | ||
| + | *[[CRISPR-Cas9 Part II]] | ||
| + | *[[CRISPR-Cas]] | ||
| + | '''Background''' | ||
| + | |||
Highlights | Highlights | ||
| - | *CRISPR-Cas9 is a powerful tool to modulate transcription in wide range of cell types. | + | *'''CRISPR-Cas9 (CRISPR type II-A)''' is a powerful tool to modulate transcription in wide range of cell types. |
*An expanding set of CRISPR-based transcription effectors is available. | *An expanding set of CRISPR-based transcription effectors is available. | ||
*Gene networks can be efficiently probed and modified for biotechnology applications.<ref name="Did">PMID:27344519</ref> | *Gene networks can be efficiently probed and modified for biotechnology applications.<ref name="Did">PMID:27344519</ref> | ||
| Line 11: | Line 17: | ||
Since the early days of genetic engineering there has been a need for control of gene expression. Naturally occurring transcription factors (TFs) have traditionally been used to achieve this goal (reviewed in <ref name="Prin1">PMID:24781324</ref>). However, their limited DNA binding sequence space required installing specific sequences within the transcription regulatory elements of the target genes. This can be technically difficult and may have unintended consequences on gene expression. Zinc fingers (ZFs) and transcription activator-like effectors (TALEs) were developed to overcome the fixed binding sequence requirements of native TFs. However, both ZFs and TALEs have significant limitations. ZFs have complicated design criteria and large highly repetitive TALE genes are difficult to synthesize and clone (reviewed in <ref name="Prin2">PMID:23718948</ref><ref name="Prin3">PMID:24584096</ref>). These challenges have recently been overcome using CRISPR-Cas9 based TFs. The biochemical properties of CRISPR-Cas9 based TFs that enable such flexibility and describe their applications to synthetic gene circuit design and multi-plexed perturbation of native gene networks.<ref name="Did">PMID:27344519</ref> | Since the early days of genetic engineering there has been a need for control of gene expression. Naturally occurring transcription factors (TFs) have traditionally been used to achieve this goal (reviewed in <ref name="Prin1">PMID:24781324</ref>). However, their limited DNA binding sequence space required installing specific sequences within the transcription regulatory elements of the target genes. This can be technically difficult and may have unintended consequences on gene expression. Zinc fingers (ZFs) and transcription activator-like effectors (TALEs) were developed to overcome the fixed binding sequence requirements of native TFs. However, both ZFs and TALEs have significant limitations. ZFs have complicated design criteria and large highly repetitive TALE genes are difficult to synthesize and clone (reviewed in <ref name="Prin2">PMID:23718948</ref><ref name="Prin3">PMID:24584096</ref>). These challenges have recently been overcome using CRISPR-Cas9 based TFs. The biochemical properties of CRISPR-Cas9 based TFs that enable such flexibility and describe their applications to synthetic gene circuit design and multi-plexed perturbation of native gene networks.<ref name="Did">PMID:27344519</ref> | ||
| - | + | ====Transcriptional regulation with CRISPR-Cas9==== | |
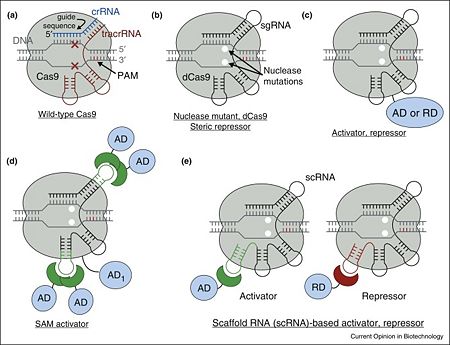
| - | + | [[Image:1-s2.0-S0958166916301537-gr1.jpg|left|450px|thumb|'''Figure 1.''' Overview of Cas9 nuclease and dCas9-based transcription factors. (a) Wild-type Cas9 endonuclease guided by crRNA:tracrRNA to a specific site in DNA creates a double-stranded DNA break. (b) dCas9, nuclease deactivated mutant of Cas9, is an RNA programmable DNA binding protein. It can act as a steric repressor of transcription in prokaryotes and eukaryotes. sgRNA is an artificial chimeric molecule consisting of crRNA and tracrRNA molecules connected with a short loop. (c) dCas9 fusion with various transcription effectors can be used to repress or activate transcription. (d) Effector domains can be recruited by sgRNA in addition to dCas9 for enhanced activity. (e) sgRNA can be modified with specific protein binding hairpins to concurrently recruit repressor or activator domains in the same cell.<ref name="Did">PMID:27344519</ref>]] | |
| + | {{Clear}} | ||
| - | + | Cas9 is a key protein of bacterial Type II CRISPR adaptive immune system (reviewed in <ref name="Prin4">PMID:26432244</ref>). In its native context, Cas9 is an RNA-guided endonuclease that is responsible for targeted degradation of the invading foreign DNA–plasmids and phages. Cas9 is directed to its DNA targets by forming a ribonucleoprotein complex with two small non-coding RNAs: CRISPR RNA (crRNA) and trans-activating crRNA (tracrRNA) (Figure 1a). In the less common class 2 CRISPR-Cas systems (types II, V, and VI), which are almost completely restricted to bacteria, the effector complex is represented by a single multidomain protein <ref name="Rev430">doi:10.1038/nrmicro3569</ref>. The best-characterized class 2 effector is Cas9 (type II), the RNA-dependent endonuclease that contains two unrelated nuclease domains, HNH and RuvC, that are responsible for the cleavage of the target and the displaced strand, respectively, in the crRNA–target DNA complex (<scene name='74/746096/Cv3/1'>Domain organization of nuclease lobe of Cas9 from S. pyogenes</scene>, [[4zt0]]). The type II loci also encode a trans-acting CRISPR RNA (tracrRNA) that evolved from the corresponding CRISPR repeat and is essential for pre-crRNA processing and target recognition in type II systems. Cas9 is directed to its DNA targets by forming a ribonucleoprotein complex with these 2 small non-coding RNAs: crRNA and tracrRNA. By elegant engineering, <scene name='74/742625/Cv3/8'>crRNA and tracrRNA can be joined end-to-end and transcribed as a single guide RNA (sgRNA)</scene> ([[4zt9]]<ref name="dCAS9">PMID:26113724</ref>) that too efficiently directs Cas9 protein to DNA targets encoded within the guide sequence of sgRNA <ref name="Jinek">PMID:22745249</ref>: | |
| - | ''Examples of 3D structures of CRISPR RNA (crRNA)'' | ||
| - | *<scene name='74/742625/Cv3/1'>CRISPR-Cas Cpf1 endonuclease-crRNA-DNA ternary complex</scene> from ''Acidaminococcus sp. BV3L6'' ([[5kk5]]). | ||
| - | *<scene name='74/742625/Cv2/10'>Crystal structure of Acidaminococcus sp. Cpf1 in complex with crRNA and target DNA</scene> ([[5b43]]). | ||
| - | *<scene name='74/742625/Cv2/9'>crRNA-dsDNA hybrid from E. coli</scene> ([[5h9f]]). | ||
| - | *<scene name='74/742625/Cv2/7'>crRNA-dsDNA hybrid and Cascade proteins from E. coli</scene> ([[4u7u]]). | ||
| - | |||
| - | Cas9 is directed to its DNA targets by forming a ribonucleoprotein complex with two small non-coding RNAs: CRISPR RNA (crRNA) and trans-activating crRNA (tracrRNA) (Figure 2a). By elegant engineering, <scene name='74/742625/Cv3/8'>crRNA and tracrRNA can be joined end-to-end and transcribed as a single guide RNA (sgRNA)</scene> (PDB entry [[4zt9]]<ref name="dCAS9">PMID:26113724</ref>) that too efficiently directs Cas9 protein to DNA targets encoded within the guide sequence of sgRNA <ref name="Jinek">PMID:22745249</ref>: | ||
| - | |||
''Examples of 3D structures of single guide RNA (sgRNA)'' | ''Examples of 3D structures of single guide RNA (sgRNA)'' | ||
*<scene name='74/742625/Cv/42'>Cas9-sgRNA-target DNA complex from Streptococcus pyogenes</scene> ([[5fw2]]). | *<scene name='74/742625/Cv/42'>Cas9-sgRNA-target DNA complex from Streptococcus pyogenes</scene> ([[5fw2]]). | ||
*<scene name='74/742625/Cv2/12'>Cas9-sgRNA-target DNA complex from Streptococcus pyogenes</scene> ([[5b2s]]). | *<scene name='74/742625/Cv2/12'>Cas9-sgRNA-target DNA complex from Streptococcus pyogenes</scene> ([[5b2s]]). | ||
*<scene name='74/742625/Cv2/13'>Cas9-sgRNA-target DNA complex from Francisella tularensis</scene> ([[5b2p]]). | *<scene name='74/742625/Cv2/13'>Cas9-sgRNA-target DNA complex from Francisella tularensis</scene> ([[5b2p]]). | ||
| - | *<scene name='74/742625/Cv3/2'>Cas9-sgRNA-target DNA complex from Staphylococcus aureus</scene> ([[4axw]]) | + | *<scene name='74/742625/Cv3/2'>Cas9-sgRNA-target DNA complex from Staphylococcus aureus</scene> ([[4axw]]). |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | The <scene name='74/742625/Cv3/4'>optimal DNA target of the complex is determined by a Watson–Crick base pairing of a short ∼20-nt sequence within sgRNA (within the crRNA in wild-type)</scene>, termed the guide sequence, adjacent to a <scene name='74/742625/Cv3/10'>few nucleotide long conserved motif recognized directly by Cas9 protein (protospacer adjacent motif, PAM)</scene> <ref name="Jinek">PMID:22745249</ref><ref name="Prin6">PMID:22949671</ref>. Despite this, a <scene name='74/742625/Cv/44'>few mismatches between guide sequence and target DNA can be tolerated</scene> <ref name="Jinek">PMID:22745249</ref><ref name="Prin7">PMID:23452860</ref><ref name="Prin8">PMID:23761437</ref><ref name="Prin9">PMID:24837660</ref>, more so within the 5’ proximal position of the guide sequence. | |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
*<scene name='74/742625/Cv4/3'>When Cas9 is in the apo state, its PAM-interacting cleft is largely disordered</scene><ref name="dCAS9">PMID:26113724</ref> ([[4cmp]]<ref name="CAS9">PMID:24505130</ref>). | *<scene name='74/742625/Cv4/3'>When Cas9 is in the apo state, its PAM-interacting cleft is largely disordered</scene><ref name="dCAS9">PMID:26113724</ref> ([[4cmp]]<ref name="CAS9">PMID:24505130</ref>). | ||
*<scene name='74/742625/Cv4/4'>Pre-target state of CAS9 with sgRNA</scene> ([[4zt0]])<ref name="dCAS9">PMID:26113724</ref>. | *<scene name='74/742625/Cv4/4'>Pre-target state of CAS9 with sgRNA</scene> ([[4zt0]])<ref name="dCAS9">PMID:26113724</ref>. | ||
| Line 142: | Line 41: | ||
*<scene name='74/742625/Cv4/15'>Cas9 interacts with both separated DNA strands</scene> (sgRNA is not shown). | *<scene name='74/742625/Cv4/15'>Cas9 interacts with both separated DNA strands</scene> (sgRNA is not shown). | ||
| - | Cas9 nuclease can be converted into <scene name='74/742625/Cv4/8'>deactivated Cas9 (dCas9)</scene> (PDB entry [[4zt9]]), an RNA-programmable DNA-binding protein, by <scene name='74/742625/Cv4/9'>mutating two key residues within its nuclease domains</scene> (Figure | + | In the type II-A system, the Cas9-tracrRNA complex and Csn2 are involved in spacer acquisition along with the Cas1-Cas2 complex <ref name="Rev453">doi:10.1101/gad.257550.114</ref><ref name="Rev471">doi:10.1038/nature14245</ref>; the involvement of Cas9 in adaptation is likely to be a general feature of type II systems. Although the key residues of Cas9 involved in PAM recognition are dispensable for spacer acquisition, they are essential for the incorporation of new spacers with the correct PAM sequence <ref name="Rev471">doi:10.1038/nature14245</ref>. The involvement of Cas9 in PAM recognition and protospacer selection <ref name="Rev471">doi:10.1038/nature14245</ref> suggests that in type II systems Cas1 may have lost this role. |
| + | |||
| + | Cas9 nuclease can be converted into <scene name='74/742625/Cv4/8'>deactivated Cas9 (dCas9)</scene> (PDB entry [[4zt9]]), an RNA-programmable DNA-binding protein, by <scene name='74/742625/Cv4/9'>mutating two key residues within its nuclease domains</scene> (Figure 1b) <ref name="dCAS9">PMID:26113724</ref><ref name="Did">PMID:27344519</ref><ref name="Jinek">PMID:22745249</ref><ref name="Prin6">PMID:22949671</ref>. | ||
In the simplest case, dCas9 can repress transcription by sterically interfering with transcription initiation or elongationby being targeted to the gene of interest with a properly chosen sgRNA <ref name="Jinek">PMID:22745249</ref><ref name="Prin6">PMID:22949671</ref><ref name="Prin7">PMID:23452860</ref><ref name="Prin8">PMID:23761437</ref><ref name="Prin10">PMID:25422271</ref><ref name="Prin11">PMID:26390083</ref><ref name="Prin12">PMID:23849981</ref><ref name="Prin13">PMID:23977949</ref><ref name="Prin14">PMID:24797424</ref>. The repression strength is strongly dependent on the position with respect to the target promoter as well as the nature of promoter itself <ref name="Prin7">PMID:23452860</ref><ref name="Prin8">PMID:23761437</ref><ref name="Prin10">PMID:25422271</ref><ref name="Prin11">PMID:26390083</ref>. In prokaryotes, repression of up to 1000-fold was achieved when targeting dCas9 to either DNA strand within a promoter or to the non-template DNA strand downstream. However, in eukaryotic cells such steric repression is weaker: only up to 2-fold and 20-fold repression was observed with natural promoters in mammalian and yeast cells correspondingly<ref name="Prin7">PMID:23452860</ref><ref name="Prin12">PMID:23849981</ref><ref name="Prin13">PMID:23977949</ref>. As a notable exception, synthetic promoters specifically constructed for direct repression by dCas9 can be repressed up to 100-fold in mammalian cells<ref name="Prin14">PMID:24797424</ref>. | In the simplest case, dCas9 can repress transcription by sterically interfering with transcription initiation or elongationby being targeted to the gene of interest with a properly chosen sgRNA <ref name="Jinek">PMID:22745249</ref><ref name="Prin6">PMID:22949671</ref><ref name="Prin7">PMID:23452860</ref><ref name="Prin8">PMID:23761437</ref><ref name="Prin10">PMID:25422271</ref><ref name="Prin11">PMID:26390083</ref><ref name="Prin12">PMID:23849981</ref><ref name="Prin13">PMID:23977949</ref><ref name="Prin14">PMID:24797424</ref>. The repression strength is strongly dependent on the position with respect to the target promoter as well as the nature of promoter itself <ref name="Prin7">PMID:23452860</ref><ref name="Prin8">PMID:23761437</ref><ref name="Prin10">PMID:25422271</ref><ref name="Prin11">PMID:26390083</ref>. In prokaryotes, repression of up to 1000-fold was achieved when targeting dCas9 to either DNA strand within a promoter or to the non-template DNA strand downstream. However, in eukaryotic cells such steric repression is weaker: only up to 2-fold and 20-fold repression was observed with natural promoters in mammalian and yeast cells correspondingly<ref name="Prin7">PMID:23452860</ref><ref name="Prin12">PMID:23849981</ref><ref name="Prin13">PMID:23977949</ref>. As a notable exception, synthetic promoters specifically constructed for direct repression by dCas9 can be repressed up to 100-fold in mammalian cells<ref name="Prin14">PMID:24797424</ref>. | ||
| - | === | + | '''Cas9-sgRNA-target DNA complexes from Streptococcus pyogenes:''' |
| + | *<scene name='74/746096/Cv3/1'>Domain organization of nuclease lobe of Cas9 from S. pyogenes</scene> ([[4zt0]]) | ||
| + | *<scene name='74/742625/Cv/42'>Cas9-sgRNA-target DNA complex from Streptococcus pyogenes</scene> ([[5fw2]]). | ||
| + | *<scene name='74/742625/Cv2/12'>Cas9-sgRNA-target DNA complex from Streptococcus pyogenes</scene> ([[5b2s]]). | ||
| + | *<scene name='74/742625/Cv4/5'>CAS9 with sgRNA and tagret DNA</scene> ([[4oo8]]). | ||
| + | *<scene name='74/742625/Cv4/11'>Cas9-sgRNA-dsDNA complex</scene> ([[5f9r]]). | ||
| + | Other representatives: [[5y36]], [[4un3]]. | ||
| - | == | + | '''For continuation please see [[CRISPR-Cas9 Part II]]''' |
| + | =See aslo= | ||
| + | *[[Cas9]] | ||
| + | *[[Endonuclease]] | ||
| + | *[[Cas9 (hebrew)]] | ||
</StructureSection> | </StructureSection> | ||
== References == | == References == | ||
<references/> | <references/> | ||
| + | |||
[[Category:Topic Page]] | [[Category:Topic Page]] | ||
[[Category:Crispr]] | [[Category:Crispr]] | ||
[[Category: Crispr-associated]] | [[Category: Crispr-associated]] | ||
[[Category: endonuclease]] | [[Category: endonuclease]] | ||
| + | [[Category: Cas9]] | ||
Current revision
| |||||||||||
References
- ↑ 1.0 1.1 1.2 1.3 1.4 Didovyk A, Borek B, Tsimring L, Hasty J. Transcriptional regulation with CRISPR-Cas9: principles, advances, and applications. Curr Opin Biotechnol. 2016 Aug;40:177-84. doi: 10.1016/j.copbio.2016.06.003. Epub, 2016 Jun 23. PMID:27344519 doi:http://dx.doi.org/10.1016/j.copbio.2016.06.003
- ↑ Brophy JA, Voigt CA. Principles of genetic circuit design. Nat Methods. 2014 May;11(5):508-20. doi: 10.1038/nmeth.2926. PMID:24781324 doi:http://dx.doi.org/10.1038/nmeth.2926
- ↑ Straubeta A, Lahaye T. Zinc fingers, TAL effectors, or Cas9-based DNA binding proteins: what's best for targeting desired genome loci? Mol Plant. 2013 Sep;6(5):1384-7. doi: 10.1093/mp/sst075. Epub 2013 May 29. PMID:23718948 doi:http://dx.doi.org/10.1093/mp/sst075
- ↑ Sander JD, Joung JK. CRISPR-Cas systems for editing, regulating and targeting genomes. Nat Biotechnol. 2014 Apr;32(4):347-55. doi: 10.1038/nbt.2842. Epub 2014 Mar 2. PMID:24584096 doi:http://dx.doi.org/10.1038/nbt.2842
- ↑ Marraffini LA. CRISPR-Cas immunity in prokaryotes. Nature. 2015 Oct 1;526(7571):55-61. doi: 10.1038/nature15386. PMID:26432244 doi:http://dx.doi.org/10.1038/nature15386
- ↑ Makarova KS, Wolf YI, Alkhnbashi OS, Costa F, Shah SA, Saunders SJ, Barrangou R, Brouns SJ, Charpentier E, Haft DH, Horvath P, Moineau S, Mojica FJ, Terns RM, Terns MP, White MF, Yakunin AF, Garrett RA, van der Oost J, Backofen R, Koonin EV. An updated evolutionary classification of CRISPR-Cas systems. Nat Rev Microbiol. 2015 Nov;13(11):722-36. doi: 10.1038/nrmicro3569. Epub 2015, Sep 28. PMID:26411297 doi:http://dx.doi.org/10.1038/nrmicro3569
- ↑ 7.0 7.1 7.2 7.3 Jiang F, Zhou K, Ma L, Gressel S, Doudna JA. STRUCTURAL BIOLOGY. A Cas9-guide RNA complex preorganized for target DNA recognition. Science. 2015 Jun 26;348(6242):1477-81. doi: 10.1126/science.aab1452. PMID:26113724 doi:http://dx.doi.org/10.1126/science.aab1452
- ↑ 8.0 8.1 8.2 8.3 8.4 Jinek M, Chylinski K, Fonfara I, Hauer M, Doudna JA, Charpentier E. A programmable dual-RNA-guided DNA endonuclease in adaptive bacterial immunity. Science. 2012 Aug 17;337(6096):816-21. doi: 10.1126/science.1225829. Epub 2012, Jun 28. PMID:22745249 doi:http://dx.doi.org/10.1126/science.1225829
- ↑ 9.0 9.1 9.2 Gasiunas G, Barrangou R, Horvath P, Siksnys V. Cas9-crRNA ribonucleoprotein complex mediates specific DNA cleavage for adaptive immunity in bacteria. Proc Natl Acad Sci U S A. 2012 Sep 25;109(39):E2579-86. Epub 2012 Sep 4. PMID:22949671 doi:http://dx.doi.org/10.1073/pnas.1208507109
- ↑ 10.0 10.1 10.2 10.3 Qi LS, Larson MH, Gilbert LA, Doudna JA, Weissman JS, Arkin AP, Lim WA. Repurposing CRISPR as an RNA-guided platform for sequence-specific control of gene expression. Cell. 2013 Feb 28;152(5):1173-83. doi: 10.1016/j.cell.2013.02.022. PMID:23452860 doi:http://dx.doi.org/10.1016/j.cell.2013.02.022
- ↑ 11.0 11.1 11.2 Bikard D, Jiang W, Samai P, Hochschild A, Zhang F, Marraffini LA. Programmable repression and activation of bacterial gene expression using an engineered CRISPR-Cas system. Nucleic Acids Res. 2013 Aug;41(15):7429-37. doi: 10.1093/nar/gkt520. Epub 2013, Jun 12. PMID:23761437 doi:http://dx.doi.org/10.1093/nar/gkt520
- ↑ Kuscu C, Arslan S, Singh R, Thorpe J, Adli M. Genome-wide analysis reveals characteristics of off-target sites bound by the Cas9 endonuclease. Nat Biotechnol. 2014 Jul;32(7):677-83. doi: 10.1038/nbt.2916. Epub 2014 May 18. PMID:24837660 doi:http://dx.doi.org/10.1038/nbt.2916
- ↑ Jinek M, Jiang F, Taylor DW, Sternberg SH, Kaya E, Ma E, Anders C, Hauer M, Zhou K, Lin S, Kaplan M, Iavarone AT, Charpentier E, Nogales E, Doudna JA. Structures of Cas9 Endonucleases Reveal RNA-Mediated Conformational Activation. Science. 2014 Feb 6. PMID:24505130 doi:http://dx.doi.org/10.1126/science.1247997
- ↑ Nishimasu H, Ran FA, Hsu PD, Konermann S, Shehata SI, Dohmae N, Ishitani R, Zhang F, Nureki O. Crystal structure of Cas9 in complex with guide RNA and target DNA. Cell. 2014 Feb 27;156(5):935-49. doi: 10.1016/j.cell.2014.02.001. Epub 2014 Feb, 13. PMID:24529477 doi:http://dx.doi.org/10.1016/j.cell.2014.02.001
- ↑ Jiang F, Taylor DW, Chen JS, Kornfeld JE, Zhou K, Thompson AJ, Nogales E, Doudna JA. Structures of a CRISPR-Cas9 R-loop complex primed for DNA cleavage. Science. 2016 Jan 14. pii: aad8282. PMID:26841432 doi:http://dx.doi.org/10.1126/science.aad8282
- ↑ Wei Y, Terns RM, Terns MP. Cas9 function and host genome sampling in Type II-A CRISPR-Cas adaptation. Genes Dev. 2015 Feb 15;29(4):356-61. doi: 10.1101/gad.257550.114. PMID:25691466 doi:http://dx.doi.org/10.1101/gad.257550.114
- ↑ 17.0 17.1 17.2 Heler R, Samai P, Modell JW, Weiner C, Goldberg GW, Bikard D, Marraffini LA. Cas9 specifies functional viral targets during CRISPR-Cas adaptation. Nature. 2015 Mar 12;519(7542):199-202. doi: 10.1038/nature14245. Epub 2015 Feb, 18. PMID:25707807 doi:http://dx.doi.org/10.1038/nature14245
- ↑ 18.0 18.1 Nielsen AA, Voigt CA. Multi-input CRISPR/Cas genetic circuits that interface host regulatory networks. Mol Syst Biol. 2014 Nov 24;10:763. doi: 10.15252/msb.20145735. PMID:25422271
- ↑ 19.0 19.1 Didovyk A, Borek B, Hasty J, Tsimring L. Orthogonal Modular Gene Repression in Escherichia coli Using Engineered CRISPR/Cas9. ACS Synth Biol. 2016 Jan 15;5(1):81-8. doi: 10.1021/acssynbio.5b00147. Epub 2015 , Sep 30. PMID:26390083 doi:http://dx.doi.org/10.1021/acssynbio.5b00147
- ↑ 20.0 20.1 Gilbert LA, Larson MH, Morsut L, Liu Z, Brar GA, Torres SE, Stern-Ginossar N, Brandman O, Whitehead EH, Doudna JA, Lim WA, Weissman JS, Qi LS. CRISPR-mediated modular RNA-guided regulation of transcription in eukaryotes. Cell. 2013 Jul 18;154(2):442-51. doi: 10.1016/j.cell.2013.06.044. Epub 2013 Jul, 11. PMID:23849981 doi:http://dx.doi.org/10.1016/j.cell.2013.06.044
- ↑ 21.0 21.1 Farzadfard F, Perli SD, Lu TK. Tunable and multifunctional eukaryotic transcription factors based on CRISPR/Cas. ACS Synth Biol. 2013 Oct 18;2(10):604-13. doi: 10.1021/sb400081r. Epub 2013 Sep, 11. PMID:23977949 doi:http://dx.doi.org/10.1021/sb400081r
- ↑ 22.0 22.1 Kiani S, Beal J, Ebrahimkhani MR, Huh J, Hall RN, Xie Z, Li Y, Weiss R. CRISPR transcriptional repression devices and layered circuits in mammalian cells. Nat Methods. 2014 Jul;11(7):723-6. doi: 10.1038/nmeth.2969. Epub 2014 May 5. PMID:24797424 doi:http://dx.doi.org/10.1038/nmeth.2969