This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Acyl carrier protein synthase
From Proteopedia
| (7 intermediate revisions not shown.) | |||
| Line 1: | Line 1: | ||
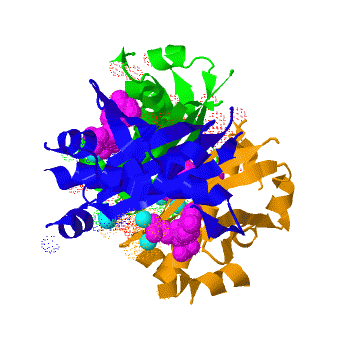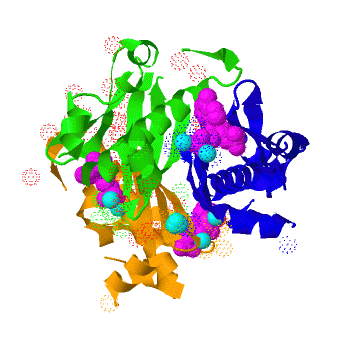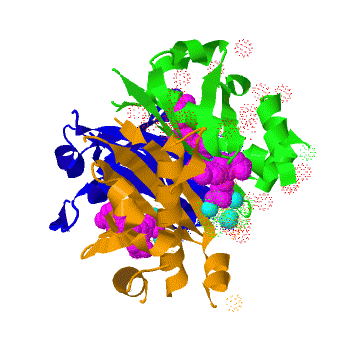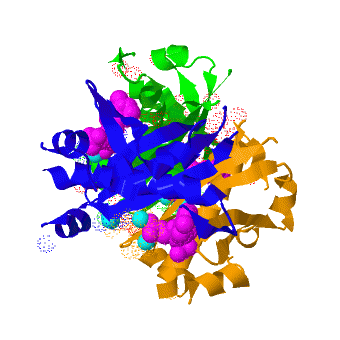
| - | <StructureSection load=' | + | <StructureSection load='' size='350' scene='3hqj/Trimer/1' caption="Acyl carrier protein synthase complex with CoA and Mg+2 ion (cyan) [[3hqj]]" > |
The crystal structure of '''Acyl carrier protein synthase (AcpS)''' from [http://http://en.wikipedia.org/wiki/Mycobacterium_tuberculosis ''Mycobacterium tuberculosis''] (''Mtb'') was solved at 1.95 Å ([[3hqj]]). It crystallized as one <scene name='3hqj/Trimer/2'>monomer</scene> per asymmetric unit. Since ''Mtb'' AcpS has biologically active trimeric arrangement, <scene name='3hqj/Trimer/3'>AcpS trimer</scene> (in <span style="color:lime;background-color:black;font-weight:bold;">green</span>, <font color='blue'><b>blue</b></font>, and <scene name='3hqj/Trimer/3'>AcpS trimer</scene> (in <span style="color:orange;background-color:black;font-weight:bold;">orange</span>) was constructed using the 3-fold crystallographic symmetry in the ''P''23 space group. | The crystal structure of '''Acyl carrier protein synthase (AcpS)''' from [http://http://en.wikipedia.org/wiki/Mycobacterium_tuberculosis ''Mycobacterium tuberculosis''] (''Mtb'') was solved at 1.95 Å ([[3hqj]]). It crystallized as one <scene name='3hqj/Trimer/2'>monomer</scene> per asymmetric unit. Since ''Mtb'' AcpS has biologically active trimeric arrangement, <scene name='3hqj/Trimer/3'>AcpS trimer</scene> (in <span style="color:lime;background-color:black;font-weight:bold;">green</span>, <font color='blue'><b>blue</b></font>, and <scene name='3hqj/Trimer/3'>AcpS trimer</scene> (in <span style="color:orange;background-color:black;font-weight:bold;">orange</span>) was constructed using the 3-fold crystallographic symmetry in the ''P''23 space group. | ||
| Line 10: | Line 10: | ||
The ''B. subtilis'' AcpS trimer ([[1f80]]) <scene name='3hqj/Acp/2'>binds</scene> three molecules of the acyl carrier protein (ASP). The interactions between ''B. subtilis'' AcpS and ACP are predominantly <scene name='3hqj/Acp/3'>electrostatic</scene>. The ''B. subtilis'' AcpS (white) is shown in spacefill representation, the agrinines, lysines, and histidines are colored <font color='blue'><b>blue</b></font>, while aspartates and glutamates are colored <font color='red'><b>red</b></font>. The ACP molecule (<span style="color:lime;background-color:black;font-weight:bold;">green</span>) is shown in ribbon representation with aspartates and glutamates as sticks and colored <font color='red'><b>red</b></font>. The ''B. subtilis'' AcpS has large <scene name='3hqj/Acp/4'>electropositive interface</scene> with ASP. <scene name='3hqj/Acp/5'>Electrostatic representation</scene> of ''Mtb'' AcpS surface using the similar orientation as ''B. subtilis'' AcpS, shows a moderate electronegative nature in the putative ACP binding site near the <font color='red'><b>ASP 15</b></font>. The ''Mtb'' ASPM structure ([[1klp]], corresponding to ACP) demonstrates considerably lower negative charge. So, the electrostatic interactions between ''Mtb'' AcpS and ASPM are, probably, less important. | The ''B. subtilis'' AcpS trimer ([[1f80]]) <scene name='3hqj/Acp/2'>binds</scene> three molecules of the acyl carrier protein (ASP). The interactions between ''B. subtilis'' AcpS and ACP are predominantly <scene name='3hqj/Acp/3'>electrostatic</scene>. The ''B. subtilis'' AcpS (white) is shown in spacefill representation, the agrinines, lysines, and histidines are colored <font color='blue'><b>blue</b></font>, while aspartates and glutamates are colored <font color='red'><b>red</b></font>. The ACP molecule (<span style="color:lime;background-color:black;font-weight:bold;">green</span>) is shown in ribbon representation with aspartates and glutamates as sticks and colored <font color='red'><b>red</b></font>. The ''B. subtilis'' AcpS has large <scene name='3hqj/Acp/4'>electropositive interface</scene> with ASP. <scene name='3hqj/Acp/5'>Electrostatic representation</scene> of ''Mtb'' AcpS surface using the similar orientation as ''B. subtilis'' AcpS, shows a moderate electronegative nature in the putative ACP binding site near the <font color='red'><b>ASP 15</b></font>. The ''Mtb'' ASPM structure ([[1klp]], corresponding to ACP) demonstrates considerably lower negative charge. So, the electrostatic interactions between ''Mtb'' AcpS and ASPM are, probably, less important. | ||
| - | PPT subtypes are the ''E. coli'' '''ACPS''' and the ''B. subtilis'' '''Sfp'''. For details on Spf see [[Molecular Playground/4'-PHOSPHOPANTETHEINYL TRANSFERASE (Sfp)]]. | + | PPT subtypes are the ''E. coli'' '''ACPS''' and the ''B. subtilis'' '''Sfp'''. For details on Spf see [[Molecular Playground/4'-PHOSPHOPANTETHEINYL TRANSFERASE (Sfp)]]. |
| - | + | ||
| - | + | ||
| - | + | *'''Beta-ketoacyl-acyl carrier protein synthase II''' is involved in temperature controlled fatty acid synthesis<ref>PMID:6988423</ref>. | |
| - | + | See also [[Acyl carrier protein]], [[Fatty acid synthesis]]. | |
| - | + | ||
| - | + | ==3D structures of acyl carrier protein synthase== | |
| - | + | [[Acyl carrier protein synthase 3D structures]] | |
| - | + | ||
| - | + | </StructureSection> | |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | *ACPS III complex | ||
| - | **[[1hnd]], [[1hnh]], [[2gyo]], [[2eft]] - EcACPS III + CoA<br /> | ||
| - | **[[1hnj]] - EcACPS III + malonyl-CoA<br /> | ||
| - | **[[2qx1]] - EcACPS III + decyl-CoA<br /> | ||
| - | **[[1mzs]], [[5bnm]], [[5bnr]], [[5bns]] - EcACPS III + inhibitor<br /> | ||
| - | **[[5bqs]] - SpACPS III + inhibitor<br /> | ||
| - | **[[4z8d]] - HiACPS III + inhibitor<br /> | ||
| - | **[[1u6s]] - MtACPS III (mutant) + lauroyl-CoA<br /> | ||
| - | **[[2qnx]], [[2qo1]] - MtACPS III + undecanoic acid<br /> | ||
| - | **[[2qo0]] - MtACPS III (mutant) + undecanoic acid<br /> | ||
| - | **[[2qnz]] - MtACPS III + decyl formate<br /> | ||
| - | **[[2qny]] - MtACPS III (mutant) + decyl formate<br /> | ||
| - | **[[1ub7]] - TtACPS III (mutant)<br /> | ||
| - | **[[3il4]] - EfACPS III + CoA – ''Enterococcus faecalis''<br /> | ||
| - | **[[3il5]] - EfACPS III + benzoic acid derivative<br /> | ||
| - | **[[3il6]] - EfACPS III (mutant) + benzoic acid derivative<br /> | ||
| - | **[[4nhd]] - VcACPS III + CoA<br /> | ||
| - | **[[4x0o]] - VcACPS III-2 + acetyl-CoA<br /> | ||
| - | **[[5kp2]] - VcACPS III-2 + octanoyl-CoA<br /> | ||
| - | }} | ||
[[Category:Topic Page]] | [[Category:Topic Page]] | ||
Current revision
| |||||||||||
References
- Parris KD, Lin L, Tam A, Mathew R, Hixon J, Stahl M, Fritz CC, Seehra J, Somers WS. Crystal structures of substrate binding to Bacillus subtilis holo-(acyl carrier protein) synthase reveal a novel trimeric arrangement of molecules resulting in three active sites. Structure. 2000 Aug 15;8(8):883-95. PMID:10997907
- Dym O, Albeck S, Peleg Y, Schwarz A, Shakked Z, Burstein Y, Zimhony O. Structure-function analysis of the acyl carrier protein synthase (AcpS) from Mycobacterium tuberculosis. J Mol Biol. 2009 Nov 6;393(4):937-50. Epub 2009 Sep 3. PMID:19733180 doi:10.1016/j.jmb.2009.08.065
Proteopedia Page Contributors and Editors (what is this?)
Categories: Topic Page | Mycobacterium tuberculosis | Albeck,S. | Burstein,Y. | Dym,O. | ISPC, Israel Structural Proteomics Center. | Peleg,Y. | Schwarz,A. | Shakked,Z. | Zimhony,O. | An/fold | Cytoplasm | Fatty acid biosynthesis | ISPC | Israel Structural Proteomics Center | Lipid synthesis | Magnesium | Metal-binding | Structural genomic | Transferase