This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Sulfatase
From Proteopedia
(Difference between revisions)
| (6 intermediate revisions not shown.) | |||
| Line 1: | Line 1: | ||
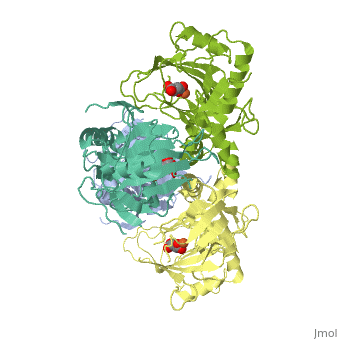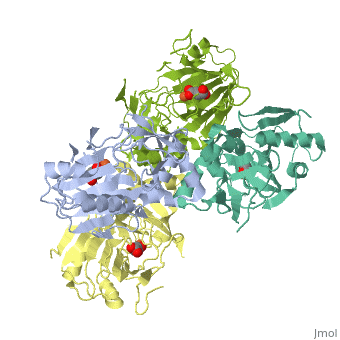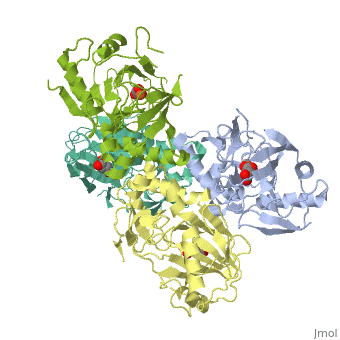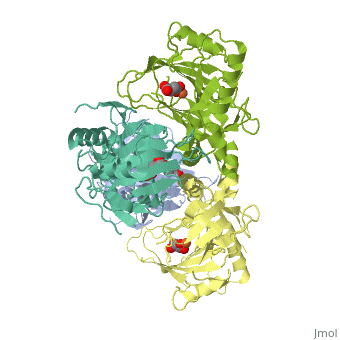
<StructureSection load='1vz4' size='340' side='right' caption='Alkylsulfatase complex with succinate and Fe+2 ion (PDB code [[1vz4]])' scene=''> | <StructureSection load='1vz4' size='340' side='right' caption='Alkylsulfatase complex with succinate and Fe+2 ion (PDB code [[1vz4]])' scene=''> | ||
| - | + | __TOC__ | |
== Function == | == Function == | ||
| Line 9: | Line 9: | ||
* '''Estrone sulfatase''' hydrolyzes sulfated steroids.<br /> | * '''Estrone sulfatase''' hydrolyzes sulfated steroids.<br /> | ||
* '''N-acetylgalactosamine-6-sulfatase''' hydrolyzes chondroitin sulfate and keratin sulfate. For details see [[Molecular Playground/Human Galactosamine-6-sulfatase]].<br /> | * '''N-acetylgalactosamine-6-sulfatase''' hydrolyzes chondroitin sulfate and keratin sulfate. For details see [[Molecular Playground/Human Galactosamine-6-sulfatase]].<br /> | ||
| + | * '''Choline sulfatase''' hydrolyzes alkyl sulfate choline-O-sulfate<ref>PMID:29458126</ref>.<br /> | ||
| + | * '''Iota-carrageenan sulfatase''' hydrolyzes sulfated polysaccharides<ref>PMID:25207269</ref>.<br /> | ||
== Disease == | == Disease == | ||
| Line 14: | Line 16: | ||
Mutations in arylsulfatase result in lysosomal storage disorders. Sulfatases are implicated in pathophysiological conditions in hormone-dependent cancer, developmental abnormalities and bacterial pathogenesis<ref>PMID:12757705</ref>. | Mutations in arylsulfatase result in lysosomal storage disorders. Sulfatases are implicated in pathophysiological conditions in hormone-dependent cancer, developmental abnormalities and bacterial pathogenesis<ref>PMID:12757705</ref>. | ||
| - | *<scene name='72/725358/Cv/ | + | *<scene name='72/725358/Cv/3'>Succinate/Fe+2 ion binding site</scene>. |
| - | + | ||
== 3D Structures of sulfatase == | == 3D Structures of sulfatase == | ||
| + | [[Sulfatase 3D structures]] | ||
| - | + | </StructureSection> | |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | **[[4fdi]] – hGalNS <br /> | ||
| - | **[[4fdj]] – hGalNS + GalNac<br /> | ||
| - | }} | ||
== References == | == References == | ||
<references/> | <references/> | ||
[[Category:Topic Page]] | [[Category:Topic Page]] | ||
Current revision
| |||||||||||
References
- ↑ Hanson SR, Best MD, Wong CH. Sulfatases: structure, mechanism, biological activity, inhibition, and synthetic utility. Angew Chem Int Ed Engl. 2004 Nov 5;43(43):5736-63. PMID:15493058 doi:http://dx.doi.org/10.1002/anie.200300632
- ↑ van Loo B, Schober M, Valkov E, Heberlein M, Bornberg-Bauer E, Faber K, Hyvonen M, Hollfelder F. Structural and Mechanistic Analysis of the Choline Sulfatase from Sinorhizobium melliloti: A Class I Sulfatase Specific for an Alkyl Sulfate Ester. J Mol Biol. 2018 Feb 16. pii: S0022-2836(18)30081-0. doi:, 10.1016/j.jmb.2018.02.010. PMID:29458126 doi:http://dx.doi.org/10.1016/j.jmb.2018.02.010
- ↑ Genicot SM, Groisillier A, Rogniaux H, Meslet-Cladiere L, Barbeyron T, Helbert W. Discovery of a novel iota carrageenan sulfatase isolated from the marine bacterium Pseudoalteromonas carrageenovora. Front Chem. 2014 Aug 26;2:67. doi: 10.3389/fchem.2014.00067. eCollection 2014. PMID:25207269 doi:http://dx.doi.org/10.3389/fchem.2014.00067
- ↑ Dierks T, Schmidt B, Borissenko LV, Peng J, Preusser A, Mariappan M, von Figura K. Multiple sulfatase deficiency is caused by mutations in the gene encoding the human C(alpha)-formylglycine generating enzyme. Cell. 2003 May 16;113(4):435-44. PMID:12757705