This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Sandbox reserved 1560
From Proteopedia
(Difference between revisions)
| (One intermediate revision not shown.) | |||
| Line 8: | Line 8: | ||
== Function == | == Function == | ||
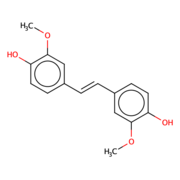
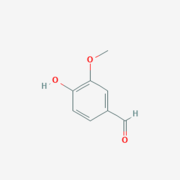
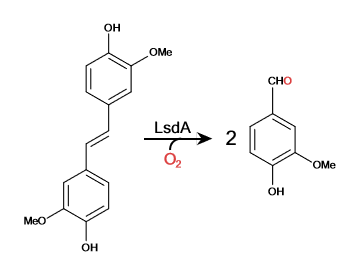
| - | Lignostilbene-α,β-dioxygenase A (LsdA) is an enzyme found in the bacteria ''Sphingomonas paucimbilis''.<ref>PMID:31292192</ref> | + | Lignostilbene-α,β-dioxygenase A (LsdA) is an enzyme found in the bacteria ''Sphingomonas paucimbilis''.<ref>PMID:31292192</ref> Lsd is a carotenoid oxygenases that involve the cleavage of carotenoids (CCOs). LsdA is used to catalyze the substrate, lignostilbene, into two vanillin molecules. Vanillin is a member of the benzaldehydes class and plays a role in the plant metabolite<ref>https://pubchem.ncbi.nlm.nih.gov/compound/Vanillin</ref>. Lignin is found in the plant cell walls, lignocellulose, and it gives the plant its rigid structure. The depolymerization of the lignin can allow for better use of the plants resources as a source of biofuel. |
===''Lignostilbene''=== | ===''Lignostilbene''=== | ||
| Line 24: | Line 24: | ||
== Structural highlights == | == Structural highlights == | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
Lignostilbene is a <scene name='83/830391/Chain_distingshin/2'>quationary</scene> structure with two identical protein chains working together. The proteins are partially composed of <scene name='83/830391/Alpha_helix/1'>alpha helix's</scene>(Blue) and the majority by <scene name='83/830391/Beta_sheet/1'>Beta-sheets</scene> indicated in red. Together the <scene name='83/830391/Secoundary_structure_together/1'>tertiary</scene> structure work to form its relationship and interactions within itself and other bio-molecular compounds found in the environment. | Lignostilbene is a <scene name='83/830391/Chain_distingshin/2'>quationary</scene> structure with two identical protein chains working together. The proteins are partially composed of <scene name='83/830391/Alpha_helix/1'>alpha helix's</scene>(Blue) and the majority by <scene name='83/830391/Beta_sheet/1'>Beta-sheets</scene> indicated in red. Together the <scene name='83/830391/Secoundary_structure_together/1'>tertiary</scene> structure work to form its relationship and interactions within itself and other bio-molecular compounds found in the environment. | ||
| - | A <scene name='82/823083/6ojttriad/1'>catalytic triad</scene> is commonly found in the active site of the protein and binds to the ligand. The triad for LsdA are identified as PHE 59, TYR 101, and LYS 134. The <scene name='83/830391/Ligand_nsl/1'>ligands</scene> are found within the two chains of the protein structure accompanied by an Iron ion. | + | A <scene name='82/823083/6ojttriad/1'>catalytic triad</scene> is commonly found in the active site of the protein and binds to the ligand. The triad for LsdA are identified as PHE 59, TYR 101, and LYS 134. The <scene name='83/830391/Ligand_nsl/1'>ligands</scene> are found within the two chains of the protein structure, accompanied by an Iron ion. |
| + | |||
| + | A unique structure of the LsdA is the <scene name='83/830391/Rainbow_7blade_beta_propeller/2'>seven-bladed β-propeller</scene> that is found very commonly in CCOs and helps form a funnel-like active site. A few amino acids that interact with the ligand in the <scene name='83/830391/Binding_site/1'>active site</scene> by hydrogen bonding are His 167 in pink, Glu 135 in red, and Phe 308 in indigo. The <scene name='83/830391/Spacefill_of_lignostilbene/1'>spacefill</scene> of the quaternary structure shows the interaction of atoms within the structure and how they fit within each other. | ||
| + | The <scene name='83/830391/Hydrophobic/1'>hydrophobic</scene> regions are shown in grey and the polar regions are in magenta. The interactions of hydrophobicity show how the structure interacts with itself as typically hydrophilic regions are found within the structure and hydrophobic regions become the outer most regions of the protein. | ||
| + | The <scene name='82/823083/Metal_binding_site/1'>metal-binding site</scene> is surrounded by histidines that allow for the Iron ion to be kept in place. The four histidines (HIS 167, HIS 218, HIS 282, and HIS 472) form a tetragonal pyramidal and found in the active site. A cap on the seven-bladed β-propeller helps protect it from the solvent. The ion is delocalized when the hydroxystilbenoid is activated by the enzyme-catalyzed deprotonation of the 4-hydroxy group. It can also form a cation when πelectron density from the scissile double bond redistributes the iron-oxy complex. forming a peroxo-substrate cation intermediate. | ||
</StructureSection> | </StructureSection> | ||
== References == | == References == | ||
<references/> | <references/> | ||
Current revision
| This Sandbox is Reserved from Aug 26 through Dec 12, 2019 for use in the course CHEM 351 Biochemistry taught by Bonnie_Hall at the Grand View University, Des Moines, USA. This reservation includes Sandbox Reserved 1556 through Sandbox Reserved 1575. |
To get started:
More help: Help:Editing |
Lignostilbene-α,β-dioxygenase A (LsdA) Catalyzation
| |||||||||||
References
- ↑ Kuatsjah E, Verstraete MM, Kobylarz MJ, Liu AKN, Murphy MEP, Eltis LD. Identification of functionally important residues and structural features in a bacterial lignostilbene dioxygenase. J Biol Chem. 2019 Jul 10. pii: RA119.009428. doi: 10.1074/jbc.RA119.009428. PMID:31292192 doi:http://dx.doi.org/10.1074/jbc.RA119.009428
- ↑ https://pubchem.ncbi.nlm.nih.gov/compound/Vanillin
- ↑ Carroll A, Somerville C. Cellulosic biofuels. Annu Rev Plant Biol. 2009;60:165-82. doi: 10.1146/annurev.arplant.043008.092125. PMID:19014348 doi:http://dx.doi.org/10.1146/annurev.arplant.043008.092125