This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Sandbox Reserved 1718
From Proteopedia
(Difference between revisions)
| (15 intermediate revisions not shown.) | |||
| Line 3: | Line 3: | ||
=Introduction= | =Introduction= | ||
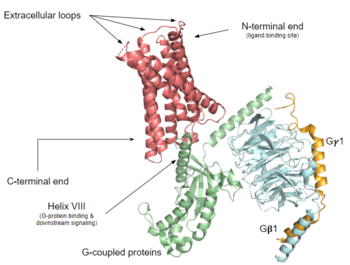
| - | [ | + | [https://proteopedia.org/wiki/index.php/G_protein-coupled_receptors G protein-coupled receptors] (GPCRs) are the largest class of integral membrane proteins.<ref name="Zhang 2015">DOI 10.14348/molcells.2015.0263</ref> GPCRs<ref name= "Zhang 2015"/><ref>PMID: 20019124</ref> are divided into five families; the [https://proteopedia.org/wiki/index.php/Sandbox_Reserved_895 rhodopsin family (class A)], the [https://proteopedia.org/wiki/index.php/4ers secretin family (class B)], the [https://proteopedia.org/wiki/index.php/6wiv glutamate family (class C)], the [https://proteopedia.org/wiki/index.php/6bd4 frizzled/taste family (class F)], and the [https://en.wikipedia.org/wiki/Adhesion_G_protein-coupled_receptor adhesion family].<ref name= "Zhang 2015"/><ref name= "Zhang 2006"/> All GPCRs contain a similar seven α-helical transmembrane domain <scene name='72/727091/Full_Structure_with_Labels/1'>(TMD)</scene> that undergoes a conformation change once bound to its ligand. This conformational change then transduces a signal to a coupled, heterotrimeric G protein which then dictates whether an intracellular signaling pathway will be initiated or inhibited. The initiation of the intracellular signaling pathway occurs in response to a variety of stimuli such as light, Ca<sup>2+</sup>, peptides, different proteins, and many more stimuli. Ultimately, intracellular signaling [https://en.wikipedia.org/wiki/G_protein%E2%80%93coupled_receptor#Physiological_roles accomplishes many interesting physiological roles].<ref name= "Zhang 2015"/><ref name= "Zhang 2006">DOI 10.1371/journal.pcbi.0020013</ref> |
| - | Human Itch G-coupled protein receptors (GPCRs), or Mast cell-related GPCRs (MRGPRX), have been identified as pruritogenic receptors and are found in human sensory neurons, specifically in the connective tissue mast cells and dorsal root ganglia in humans.<ref name= "davidson2011">DOI: 10.1016/j.tins.2010.09.002< | + | Human Itch G-coupled protein receptors (GPCRs), or Mast cell-related GPCRs (MRGPRX), have been identified as pruritogenic receptors and are found in human sensory neurons, specifically in the connective tissue mast cells and dorsal root ganglia in humans.<ref name= "davidson2011">DOI: 10.1016/j.tins.2010.09.002</ref> They are classified as class A GPCRs, however, MRGPRX receptors respond to a diverse number of agonists, antagonists, and inverse agonists some of which are not typical ligands of class A receptors. MRGPRX are involved in host defense, pseudo-allergic reactions, non-histaminergic itch, periodontitis, neurogenic inflammation, and inflammatory pain.<ref name= "davidson2011"/> |
The determination of the first structures of a ligand-activated GPCR was achieved by Robert J. Lefkowitz and Brian K. Kobilka which won them the 2012 Nobel Prize in Chemistry. They also successfully captured images of the first activated GPCR in a complex with a G protein. See [https://proteopedia.org/wiki/index.php/Nobel_Prizes_for_3D_Molecular_Structure Nobel Prizes for 3D Molecular Structure]. | The determination of the first structures of a ligand-activated GPCR was achieved by Robert J. Lefkowitz and Brian K. Kobilka which won them the 2012 Nobel Prize in Chemistry. They also successfully captured images of the first activated GPCR in a complex with a G protein. See [https://proteopedia.org/wiki/index.php/Nobel_Prizes_for_3D_Molecular_Structure Nobel Prizes for 3D Molecular Structure]. | ||
| - | == Related Enzymes == | + | ==Related Enzymes== |
| - | Among Human Itch GPCRs, MRGPRX2 is a class A GPCR that regulates mast cell degranulation and itch-related hypersensitivity reactions.<ref name="Can">DOI: 10.1038/s41586-021-04126-6</ref><ref name="Yang"/> MRGPRX2 is also a target of morphinan alkaloids, like morphine, codeine, and dextromethorphan.<ref name="Can"/><ref name="Yang"/> MRGPRX2 couples to nearly all G-protein families and subtypes with robust coupling to G<sub>q</sub> and G<sub>i</sub> families.<ref name="Can"/><ref name="Yang"/> | + | Among Human Itch GPCRs, <scene name='90/904324/Mrgprx2_receptor/4'>MRGPRX2</scene> is a class A GPCR that regulates mast cell degranulation and itch-related hypersensitivity reactions.<ref name="Can">DOI: 10.1038/s41586-021-04126-6</ref><ref name="Yang"/> MRGPRX2 is also a target of morphinan alkaloids, like morphine, codeine, and dextromethorphan.<ref name="Can"/><ref name="Yang"/> MRGPRX2 couples to nearly all G-protein families and subtypes with robust coupling to G<sub>q</sub> and G<sub>i</sub> families.<ref name="Can"/><ref name="Yang"/> |
<scene name='90/904324/7s8p_mx4/2'>MRGPRX4</scene> is another sub-group of the MRGPRX family, which mediates cholestatic itch and is a target of nateglinide drugs.<ref name="Can"/><ref name="Yang"/> MRGPRX4 also couples to G<sub>q</sub> and G<sub>i</sub> similar to MRGPRX2. | <scene name='90/904324/7s8p_mx4/2'>MRGPRX4</scene> is another sub-group of the MRGPRX family, which mediates cholestatic itch and is a target of nateglinide drugs.<ref name="Can"/><ref name="Yang"/> MRGPRX4 also couples to G<sub>q</sub> and G<sub>i</sub> similar to MRGPRX2. | ||
| Line 36: | Line 36: | ||
== Ligand Binding Site == | == Ligand Binding Site == | ||
| + | preface | ||
| + | <scene name='90/904324/Active_site_residues/3'>Sub-pocket 1</scene> | ||
<jmol> | <jmol> | ||
| Line 45: | Line 47: | ||
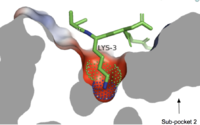
==== Sub-pocket 1 ==== | ==== Sub-pocket 1 ==== | ||
[[Image:Subpocket1.jpg.png|200px|right|thumb|Cross-sectional view of electrostatic surface of MRPRX2 sub-pocket 1 interaction with lysine 3 of cortistatin-1]] | [[Image:Subpocket1.jpg.png|200px|right|thumb|Cross-sectional view of electrostatic surface of MRPRX2 sub-pocket 1 interaction with lysine 3 of cortistatin-1]] | ||
| - | Sub-pocket 1 is formed by TM3, TM6, and ECL2.<ref name="Can"/> This sub-pocket is both small and deep which results in the binding of only a single amino acid residue, namely arginine or sometimes lysine.<ref name="Can"/> The binding is mediated by two | + | Sub-pocket 1 is formed by TM3, TM6, and ECL2.<ref name="Can"/> This sub-pocket is both small and deep which results in the binding of only a single amino acid residue, namely arginine or sometimes lysine.<ref name="Can"/> The binding is mediated by two key residues on the MRGPRX2 protein within the binding site: Glu164 and Asp184.<ref name="Can"/> The strong charge interactions of these two residues create a highly negatively charged electrostatic interaction within this sub-pocket.<ref name="Can"/> |
==== Sub-pocket 2 ==== | ==== Sub-pocket 2 ==== | ||
| Line 57: | Line 59: | ||
===Toggle Switch=== | ===Toggle Switch=== | ||
| - | The | + | The toggle switch of class A GPCRs enables the receptor to initiate the signaling cascade. However, MRGPRX2 does not contain the conserved <scene name='90/904324/Activation_mechanism/5'>toggle switch</scene> Trp. Instead, it is replaced by Gly.<ref name="Can"/> Therefore, the main residues of this motif in MRGPRX2 are Gly236, Tyr113, Phe239, and Trp243.<ref name="Can"/> As a result, TM6 is shifted closer to TM3 on the extracellular side of the membrane. This conformational change may account for the lack of ligand binding of MRGPRX2 as compared to family A receptors.<ref name="Can"/> This toggle switch swap also means that ligands, such as (R)-zinc-3573 and Cortistatin-14, bind in a different spot than ligands do on other class A GPCRs. In MRGPRX2, Gly236 is located closer to the bottom of the interface, which is the same in MRGPRX4 (Gly229). Compared with other structures, such as [https://www.rcsb.org/structure/6WHA 5-HT<sub>2A</sub>R], [https://www.rcsb.org/structure/5G53 A<sub>2A</sub>R], and [https://www.rcsb.org/structure/3SN6 β<sub>2</sub>AR], the TM6 helices of MRGPRX2 and MRGPRX4 are closer to the TM3 helix which makes the binding pocket more occluded than seen in canonical structures.<ref name="Can"/> |
===PIF/LLF motif=== | ===PIF/LLF motif=== | ||
| Line 89: | Line 91: | ||
= Clinical Relevance = | = Clinical Relevance = | ||
| - | MRGPRX2 initiates IgE-mediated anaphylactic reactions.<ref name="porebski">DOI: 10.3389/fimmu.2018.03027</ref> MRGPRX2-mediated anaphylactic responses occur more quickly than IgE-mediated responses, but the responses also tended to be more transient.<ref name="porebski"/> Common commercial drugs, like icatibant and cetrorelix, as well as neuromuscular blocking agents activate mast cells through the MRGPRX2 pathway. | + | <scene name='90/904324/Mrgprx2_receptor/4'>MRGPRX2</scene> initiates IgE-mediated anaphylactic reactions.<ref name="porebski">DOI: 10.3389/fimmu.2018.03027</ref> MRGPRX2-mediated anaphylactic responses occur more quickly than IgE-mediated responses, but the responses also tended to be more transient.<ref name="porebski"/> Common commercial drugs, like icatibant and cetrorelix, as well as neuromuscular blocking agents activate mast cells through the MRGPRX2 pathway. |
Many mutations also affect the actions of MRGPRX2. For example, a single residue mutation in sub-pocket 1 (Glu164Arg) prevented interactions between the receptor and ligands like C48/80.<ref name="porebski"/> In addition, single nucleotide polymorphisms (SNPs) have been linked to many variations of MRGPRX2 which predispose patients to hyperactivation of the receptors. Two of the most common SNPs are Asn62Thr which affects the cytoplasmic domain and Asn16His which affects the extracellular domain.<ref name="porebski"/> These mutations have been theorized to potentially protect patients from drug-induced mast cell degranulation and hypersensitivity reactions. | Many mutations also affect the actions of MRGPRX2. For example, a single residue mutation in sub-pocket 1 (Glu164Arg) prevented interactions between the receptor and ligands like C48/80.<ref name="porebski"/> In addition, single nucleotide polymorphisms (SNPs) have been linked to many variations of MRGPRX2 which predispose patients to hyperactivation of the receptors. Two of the most common SNPs are Asn62Thr which affects the cytoplasmic domain and Asn16His which affects the extracellular domain.<ref name="porebski"/> These mutations have been theorized to potentially protect patients from drug-induced mast cell degranulation and hypersensitivity reactions. | ||
| Line 100: | Line 102: | ||
*[[Pharmaceutical Drugs]] | *[[Pharmaceutical Drugs]] | ||
*[[Nobel Prizes for 3D Molecular Structure]] | *[[Nobel Prizes for 3D Molecular Structure]] | ||
| - | + | *[[Sandbox Reserved 895| Rhodopsin Family GPCRs]] | |
| + | *[[4ers| Secretin Family GPCRs]] | ||
| + | *[[6wiv| Glutamate Family GPCRs]] | ||
| + | *[[6bd4| Frizzled/Taste Family GPCRs]] | ||
| + | *[https://en.wikipedia.org/wiki/Adhesion_G_protein-coupled_receptor Adhesion Family GPCRs] | ||
Current revision
Human Itch G-Coupled Protein Receptors
| |||||||||||
Student contributors
Madeline Beck Joey Gareis