Sand box 326
From Proteopedia
| (2 intermediate revisions not shown.) | |||
| Line 41: | Line 41: | ||
Using SwissDock, it was determined that larger, cyclic molecules are good substrates that would have the best binding affinity to 3CBW, each had about a -7 kcal/mol binding affinity. These large molecules are 4-Nitrophenyl N-acetyl-β-D-glucosaminide, 4-Nitrophenyl α-D-glucopyranoside, and p-nitrophenyl phosphate. | Using SwissDock, it was determined that larger, cyclic molecules are good substrates that would have the best binding affinity to 3CBW, each had about a -7 kcal/mol binding affinity. These large molecules are 4-Nitrophenyl N-acetyl-β-D-glucosaminide, 4-Nitrophenyl α-D-glucopyranoside, and p-nitrophenyl phosphate. | ||
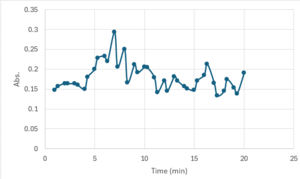
| + | == Bradford Assay Results == | ||
| + | |||
| + | 3CBW is catalytically active at a higher temperature (70 ºC) when reacted with p-nitrophenol(PNP). | ||
| + | [[Image:Screenshot_2025-04-28_110316.png |300px|center|thumb|]] | ||
== Theoretical Functionality and Proposed Bodily Purpose == | == Theoretical Functionality and Proposed Bodily Purpose == | ||
| Line 58: | Line 62: | ||
A) Dhawan, S.; Kaur, J.; Microbial Mannanases: An Overview of Production and Applications. Critical Reviews in Biotechnology 2007, 27, 197-216. DOI: 10.1080/07388550701775919 | A) Dhawan, S.; Kaur, J.; Microbial Mannanases: An Overview of Production and Applications. Critical Reviews in Biotechnology 2007, 27, 197-216. DOI: 10.1080/07388550701775919 | ||
| + | |||
B) Soni, H.; Rawat, H. K.; Pletschke, B. I.; Kango, N. Purification and characterization of Beta-mannanase from Aspergillus terreus and its applicability in depolymerization of mannans and saccharification of lignocellulosic biomass. Biotech 2016, 6, 136. DOI: 10.1007/s13205-016-0454-2 | B) Soni, H.; Rawat, H. K.; Pletschke, B. I.; Kango, N. Purification and characterization of Beta-mannanase from Aspergillus terreus and its applicability in depolymerization of mannans and saccharification of lignocellulosic biomass. Biotech 2016, 6, 136. DOI: 10.1007/s13205-016-0454-2 | ||
| + | |||
C) Cheng, L.; Duan, S.; Feng, X.; Zheng, K.; Yang, Q.; Liu, Z. Purification and Characterization of a Thermostable Beta-Mannanase from Bacillus subtilis BE-91: Potential Application in Inflammatory Diseases. BioMed Research International 2016, 2016, 1-7. DOI: 10.1155/2016/6380147 | C) Cheng, L.; Duan, S.; Feng, X.; Zheng, K.; Yang, Q.; Liu, Z. Purification and Characterization of a Thermostable Beta-Mannanase from Bacillus subtilis BE-91: Potential Application in Inflammatory Diseases. BioMed Research International 2016, 2016, 1-7. DOI: 10.1155/2016/6380147 | ||
<references/> | <references/> | ||
Current revision
3CBW Structure and Proposed Functionality
(NOTE TO ALL EDITORS: This page is part of a final project for a biochemistry lab at Elizabethtown College. Please do not edit this.)
3CBW is a homodimeric protein complex that originates from the bacterial species Bacillus Subtilis and has a mass of 80.65 kDa. It is a member of the Glycoside Hydrolase super family with structural and sequential similarities to hydrolases and mannanases. Current evidence suggests it causes the hydrolysis of glycosidic linkages via a Leu. 176 or Ile. 275 active site.
| |||||||||||
References
A) Dhawan, S.; Kaur, J.; Microbial Mannanases: An Overview of Production and Applications. Critical Reviews in Biotechnology 2007, 27, 197-216. DOI: 10.1080/07388550701775919
B) Soni, H.; Rawat, H. K.; Pletschke, B. I.; Kango, N. Purification and characterization of Beta-mannanase from Aspergillus terreus and its applicability in depolymerization of mannans and saccharification of lignocellulosic biomass. Biotech 2016, 6, 136. DOI: 10.1007/s13205-016-0454-2
C) Cheng, L.; Duan, S.; Feng, X.; Zheng, K.; Yang, Q.; Liu, Z. Purification and Characterization of a Thermostable Beta-Mannanase from Bacillus subtilis BE-91: Potential Application in Inflammatory Diseases. BioMed Research International 2016, 2016, 1-7. DOI: 10.1155/2016/6380147